Icotinib Hydrochloride |
| Catalog No.GC16244 |
An EGFR inhibitor
Products are for research use only. Not for human use. We do not sell to patients.
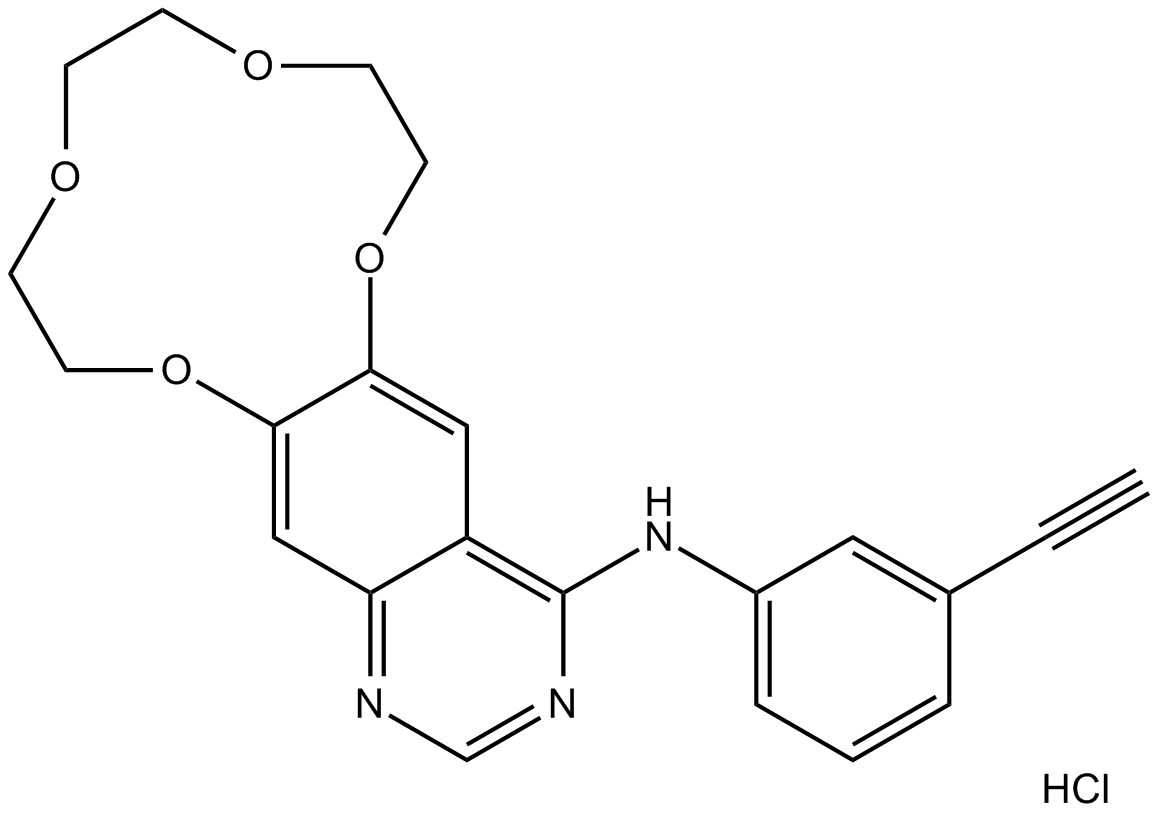
Cas No.: 1204313-51-8
Sample solution is provided at 25 µL, 10mM.
Icotinib Hydrochloride is a potent and highly selective inhibitor of epidermal growth factor receptor tyrosine kinases (EGFR-TKI) with IC50 value of 5 nM.
EGFR is an oncogenic driver which is expressed on the cell surface of normal cells and cancer cells [1], and patients with somatic mutations, particularly an exon 19 deletion or exon 21 L858R mutation, within the tyrosine kinase domain have activating mutations that lead to unchecked cell proliferation.[2] Overexpression of EGFR caused inappropriate activation of the anti-apoptotic Ras signaling pathway, found in many different types of cancer.[3]
Icotinib is a quinazoline derivative that binds reversibly to the ATP binding site of the EGFR protein, preventing completion of the signal transduction cascade.[4] Icotinib inhibited the intracellular phosphorylation of tyrosine kinase(TK) associated with the epidermal growth factor receptor (EGFR).[5]
Icotinib is indicated for the treatment for EGFR mutation-positive, advanced or metastatic non-small cell lung cancer (NSCLC) as a second-line or third-line treatment, for patients who have failed at least one prior treatment with platinum-based chemotherapy.[6]
References:
1.A Douglas Laird,and Julie M Cherrington. Small molecule tyrosine kinase inhibitors: clinical development of anticancer agents. 2003, 12(1): 51-64.
2.Raymond E, Faivre S, Armand JP: Epidermal growth factor receptor tyrosine kinase as a target for anticancer therapy. Drugs. 2000;60 Suppl 1:15-23.
3.Sordella,R. "Gefitinib-Sensitizing EGFR Mutations in Lung Cancer Activate Anti-Apoptotic Pathways". Science ,2004, 305 (5687): 1163-1167.
4.Bulgaru AM. et al. Erlotinib (Tarceva): a promising drug targeting epidermal growth factor receptor tyrosine kinase. Expert Rev Anticancer Ther. 2003 Jun;3(3):269-79.
5.Dudek AZ.et al. Skin rash and bronchoalveolar histology correlates with clinical benefit in patients treated with gefitinib as a therapy for previously treated advanced or metastatic non-small cell lung cancer. Lung Cancer. 2006, 51(1):89-96.
6.C Delbaldo, S Faivre, E Raymond. Les inhibiteurs des récepteurs de l'Epidermal Growth Factor (EGF) Epidermal growth factor inhibitors. La Revue de Médecine Interne. 2003,24(6): 372–383.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















