(2R)-Octyl-α-hydroxyglutarate ((2R)-Octyl-2-HG) (Synonyms: (2R)Octyl2-HG) |
| Catalog No.GC32754 |
(2R)-Octyl-α-hydroxyglutarate ((2R)-Octyl-2-HG) ((2R)-Octyl-2-HG) is a modified form of D-isomer 2-Hydroxyglutarate.
Products are for research use only. Not for human use. We do not sell to patients.
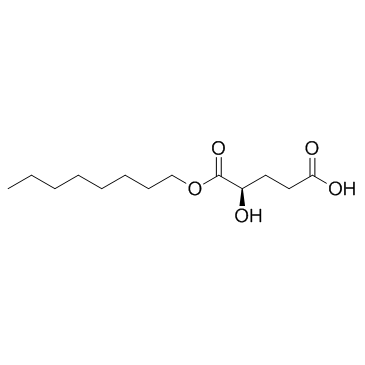
Cas No.: 1391194-67-4
Sample solution is provided at 25 µL, 10mM.
(2R)-Octyl-α-hydroxyglutarate ((2R)-Octyl-2-HG) is a modified form of D-isomer 2-Hydroxyglutarate.
Treatment of IDH1-WT HeLa cells with (2R)-Octyl-α-hydroxyglutarate, a modified form of 2-Hydroxyglutarate with high cellular uptake, results in a dose-dependent increase in DSBs in log-phase cells. It has been used to examine the contribution of D-2-Hydroxyglutarate to the oxidative mitochondrial processes of IDH1-mutated cancer cells[1].
[1]. Sulkowski PL, et al. 2-Hydroxyglutarate produced by neomorphic IDH mutations suppresses homologousrecombination and induces PARP inhibitor sensitivity. Sci Transl Med. 2017 Feb 1;9(375).
Average Rating: 5 (Based on Reviews and 12 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















