7-Aminoactinomycin D (7-AAD) (Synonyms: 7-AAD, NSC 239759) |
| Catalog No.GC33577 |
7-Aminoactinomycin D (7-AAD) (7-AAD) a fluorescent DNA stain, is a potent RNA polymerase inhibitor.
Products are for research use only. Not for human use. We do not sell to patients.
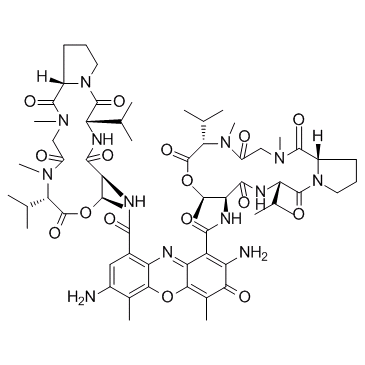
Cas No.: 7240-37-1
Sample solution is provided at 25 µL, 10mM.
Product has been cited by 1 publications
Product Documents
Quality Control & SDS
- View current batch:
- Purity: >95.00%
- COA (Certificate Of Analysis)
- SDS (Safety Data Sheet)
- Datasheet
7-Aminoactinomycin D is a fluorescent DNA stain.
[1]. Wadkins RM, et al. Actinomycin D and 7-aminoactinomycin D binding to single-stranded DNA. Biochemistry. 1991 Oct 1;30(39):9469-78.
| Cas No. | 7240-37-1 | SDF | |
| Synonyms | 7-AAD, NSC 239759 | ||
| Canonical SMILES | O=C(N[C@H](C(N1[C@@](C(N(C)CC(N(C)[C@@H](C(C)C)C(O[C@@H]2C)=O)=O)=O)([H])CCC1)=O)C(C)C)[C@H]2NC(C(C(C(OC3=C(C)C(N)=C4)=C5C)=NC3=C4C(N[C@@H]6C(N[C@H](C(N7[C@@](C(N(C)CC(N(C)[C@@H](C(C)C)C(O[C@@H]6C)=O)=O)=O)([H])CCC7)=O)C(C)C)=O)=O)=C(N)C5=O)=O | ||
| Formula | C62H87N13O16 | M.Wt | 1270.43 |
| Solubility | DMF: 10 mg/ml,DMF:PBS(pH 7.2)(1:1): 0.5 mg/ml,DMSO: 5 mg/ml | Storage | -20°C, protect from light,unstable in solution, ready to use. |
| General tips | Please select the appropriate solvent to prepare the stock solution according to the
solubility of the product in different solvents; once the solution is prepared, please store it in
separate packages to avoid product failure caused by repeated freezing and thawing.Storage method
and period of the stock solution: When stored at -80°C, please use it within 6 months; when stored
at -20°C, please use it within 1 month. To increase solubility, heat the tube to 37°C and then oscillate in an ultrasonic bath for some time. |
||
| Shipping Condition | Evaluation sample solution: shipped with blue ice. All other sizes available: with RT, or with Blue Ice upon request. | ||
Complete Stock Solution Preparation Table
| Prepare stock solution | |||

|
1 mg | 5 mg | 10 mg |
| 1 mM | 0.7871 mL | 3.9357 mL | 7.8714 mL |
| 5 mM | 0.1574 mL | 0.7871 mL | 1.5743 mL |
| 10 mM | 0.0787 mL | 0.3936 mL | 0.7871 mL |
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
 g
g
 μL
μL

Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such as vortex, ultrasound or hot water bath can be used to aid dissolving.
3. All of the above co-solvents are available for purchase on the GlpBio website.
Related Products
- GC36396 KPLH1130
- GC19304 R1487 Hydrochloride
- GC11063 BMX-IN-1
- GN10490 Olaquindox
- GC17126 EX-527 R-enantiomer
- GC35321 Aminomalonic acid
- GC14592 KW 2449
- GC34730 PROTAC CRABP-II Degrader-1
- GC16587 Nimodipine
- GC19111 CP671305
- GC43902 Indole-3-amidoxime
- GC36868 Pennogenin 3-O-beta-chacotrioside
- GC35502 Betamethasone acibutate
- GC32250 Chebulinic acid
- GC42700 Acetyl Pentapeptide-1
- GC41040 JWH 073 6-hydroxyindole metabolite
Reviews
Average Rating: 5 ★★★★★ (Based on Reviews and 12 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *


