β-Amyloid 31-35 |
| Catalog No.GC31179 |
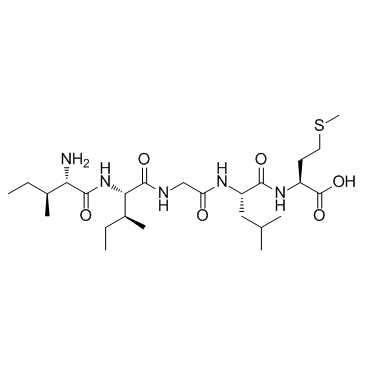
β-Amyloid 31-35 is the shortest sequence of native Amyloid-β peptide that retains neurotoxic activity.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 149385-65-9
Sample solution is provided at 25 µL, 10mM.
β-Amyloid (31-35) is the shortest sequence of native Amyloid-β peptide that retains neurotoxic activity.
β-Amyloid (31-35) is a functional cytotoxic domain of Aβ peptide. β-Amyloid (31-35) increases the phosphorylation of biotinylated Aβ(1-40), enhances CDK-1 activity, and also inhibits binding of Aβ to cyclin B1. β-Amyloid (31-35) is cytotoxic, and such an effect can be inhibited by olomoucine in differentiated human teratocarcinoma cell line, Ntera 2/cl-D1 (NT-2) neurons[1]. β-Amyloid (31-35) shows cytotoxic effect on cerebellar granule cells (CGC). β-Amyloid (31-35) also increases caspase-3 activity in a time-dependent manner (4-24 h) at 40 μM[2].
[1]. Milton NG, et al. The amyloid-beta peptide binds to cyclin B1 and increases human cyclin-dependent kinase-1 activity. Neurosci Lett. 2002 Apr 5;322(2):131-3. [2]. Misiti F, et al. Fragment 31-35 of beta-amyloid peptide induces neurodegeneration in rat cerebellar granule cells via bax gene expression and caspase-3 activation. A crucial role for the redox state of methionine-35 residue. Neurochem Int. 2006 Oct;49(5):525-32.
Average Rating: 5 (Based on Reviews and 31 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















