C11 BODIPY 581/591
|
| Catalog No.GC40165 |
C11-BODIPY581/591 is a fluorescent ratio-probe of lipid oxidation.
Products are for research use only. Not for human use. We do not sell to patients.
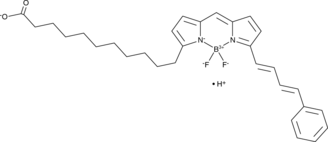
Cas No.: 217075-36-0
Sample solution is provided at 25 µL, 10mM.
- Brain Res Bull 174 (2021): 250-259.PMID:34171402
- Acta Pharm Sin B 12.2 (2022): 692-707.PMID:35256940
- J Leukocyte Biol 110.2 (2021): 301-314.PMID:34318944
- Anal Chem 93.34 (2021): 11758-11764.PMID:34410685
- ACS Appl Mater Inter 13.45 (2021): 53671.PMID:34730938
- Front Mol Biosci 8 (2021): 725274.PMID:34568428
- Oxid Med Cell Longev 2021 (2021).PMID:34970416
- Eur J Pharmacol 919 (2022): 174797.PMID:35122867
- ACS nano 16.3 (2022): 3647-3663.PMID:35266697
- Transl Cancer Res 11.5 (2022): 1173.PMID:35706810
- J Nanobiotechnol 20.1 (2022): 230.PMID:35568865
- Chin Med-UK 17.1 (2022): 82.PMID:35787281
- Biomed Pharmacother 154 (2022): 113572.PMID:35988428
- Biochem Bioph Res Co 627 (2022): 103-110.PMID:36030651
- Life Sci 308 (2022): 120925.PMID:36057399
- Ebiomedicine 84 (2022).PMID:36137413
- Plos one 17.11 (2022): e0276083.PMID:36355759
- Cells 11.22 (2022): 3580.PMID:36429010
- Eur J Pharm Sci 181 (2023): 106350..PMID:36496165
- Redox Biol 59 (2023): 102564.PMID:36473315
- Cancer Lett 554 (2023): 216033.PMID:36493901
- Sci Total Environ 855 (2023): 158715.PMID:36113792
- J Exp Clin Canc Res 42.1 (2023): 1-19.PMID:36604718
- Chem Eng J 454 (2023): 140044.
- Free Radical Bio Med 195 (2023): 329-342.PMID:36610561
- Molecules 28.8 (2023): 3389.PMID:37110625
- Chem Eng J 466 (2023): 143012.
- Free Radical Bio Med 204 (2023): 226-242.PMID:37146698
- Acta Biomater (2023).PMID:37156432
- Chem Res Chinese U (2023): 1-6.
- Biochem Pharmacol 213 (2023): 115592.PMID:37196680
- J Nanobiotechnol 21.1 (2023): 1-19.PMID:37316835
- Int J Mol Med 52.5 (2023): 1-14.PMID:37800609
- Biochem Cell Biol 100.5 (2022): 378-386.PMID:35785548
- Cell Death Discov 8.1 (2022): 431.PMID:36307402
- Eur J Neurosci 58.1 (2023): 2194-2214.PMID:37161649
- Front Cell Neurosci 16 (2022): 774297.PMID:35431808
- Front Mol Biosci 9 (2022): 860525.PMID:35860356
- Front Oncol 11 (2022): 802124.PMID:35127512
- J Nanobiotechnol 20.1 (2022): 1-15.PMID:36109814
- J Biochem Mol Toxic (2023): e23542.PMID:37712196
- Method Protocol (2023): 165-178.PMID:37578705
- Eur J Neurosci 58.1 (2023): 2194-2214.PMID:37161649
- Exp Ther Med 26.5 (2023): 1-15.PMID:37869642
- J Pharm Anal (2023).
- The Prostate (2023).PMID:37750290
- Int Immunopharmacol 127 (2024):111377.PMID:38104369
- Redox Rep 29.1 (2024):2290864.PMID:38149613
- Bioact Mater 33 (2024):483-496.PMID:38125638
- Biochem Bioph Res Co 690 (2024):149274.PMID:37995455
- J Mater Chem B (2024).PMID:38063821
- Biol Trace Elem Res (2024):1-11.PMID:38183555
- J Orthop Transl 44 (2024):114-124.
- J Mol Struct (2024):137668.
- Cell Signal (2024):111108.PMID:38369266
- J Funct Foods 113 (2024):106018.
- Kaohsiung J Med Sci (2024).PMID:38340032
- Pharm Biol 62.1 (2024):195-206.PMID:38339810
- Research (2024).PMID:38371274
- Cell Death Differ (2024):1-16.PMID:38418695
- Carcinogenesis (2024):bgae016.PMID:38422369
- Int J Biol Sci 20.5 (2024):1707.PMID:38481805
- bioRxiv (2024):2024-03.
- Sci Total Environ (2024):171818.PMID:38508245
- Appl Organomet Chem (2024):e7456.
- Heliyon (2024).
- Antioxid Redox Sign (2024).PMID:38545792
- Biomed Pharmacother 174 (2024):116542.PMID:38574620
- Bone Res 12.1 (2024):20.PMID:38553442
- Cancer Lett (2024):216826.PMID:38574881
- Heliyon (2024).PMID:38576562
C11-BODIPY581/591 is an oxidation-sensitive fluorescent fatty acid analogue with fluorescent properties in the red range of the visible spectrum (emission maximum 595 nm), allowing its application in fluorescence microscopy. C11-BODIPY581/591 is easily incorporating into membranes and fluoresces red in the intact state but shifts to green upon free radical-induced oxidation. This characteristic is highly advantageous, it makes the ratio-imaging of oxidant activities at the (sub)cellular level feasible. In addition, the fluorescent properties of C11-BODIPY581/591 allow the use of this probe in fast- and medium- throughput screening of antioxidants in living cells and model membranes in a multiwell/fluorescence reader approach[1][2].
The wavelengths of maximal excitation and emission of fluorophore C11-BODIPY581/591 corresponded to 581 and 591 nm, respectively. Addition of CumOOH/hemin, as an initiator of lipid oxidation, shifted the excitation and emission spectra to shorter wavelengths corresponding to green fluorescence (peak excitation 500 nm, emission 510 nm). C11-BODIPY581/591 is also easily oxidized by other hydroxy-, peroxy- and oxy-radical generating systems such as hydrogen peroxide/Fe2+ and 2,2’-azobis. However, this probe is relatively insensitive to SIN-1, which generates nitric oxide and superoxide[3]
References:
[1]. Drummen GP, et al. C11-BODIPY581/591, an oxidation-sensitive fluorescent lipid peroxidation probe: (micro)spectroscopic characterization and validation of methodology. Free Radic Biol Med. 2002 Aug 15;33(4):473-90.
[2]. Partyka A, et al. Detection of lipid peroxidation in frozen-thawed avian spermatozoa using C11-BODIPY581/591. Theriogenology. 2011 Jun;75(9):1623-9.
[3]. Pap EH, et al. Ratio-fluorescence microscopy of lipid oxidation in living cells using C11-BODIPY581/591. FEBS Lett. 1999 Jun 25;453(3):278-82.
Average Rating: 5 (Based on Reviews and 33 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *





