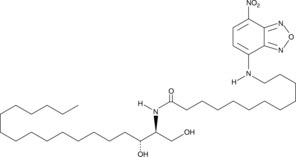
C12 NBD dihydro Ceramide (d18:0/12:0) (Synonyms: NBD Ceramide (d18:0/12:0)) |
| Catalog No.GC43011 |
C12 NBD dihydro Ceramide (d18:0/12:0) is a fluorescent ceramidase substrate.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 474943-05-0
Sample solution is provided at 25 µL, 10mM.
Ceramidase catalyzes the hydrolysis of the N-acyl linkage between the fatty acid and sphingosine base in ceramide to produce sphingosine and a free fatty acid. Three isoforms of ceramidases (acid, neutral, and alkaline) have been characterized based on differences in their catalytic pH optimum.[1][2] C12 NBD dihydro ceramide is a fluorescent ceramide analog that contains a saturated bond in the C-4/C-5 position of the sphingosine backbone. C-12 NBD ceramide, which contains the C-4/C-5 double bond, is a fluorescent ceramidase substrate that can be used for the measurement of alkaline and neutral ceramidase activity from a variety of sources.[3] C12 NBD dihydro ceramide may exhibit reduced activity in ceramidase assays compared to C-12 NBD ceramide. This is based on the observation that saturation of the C-4/C-5 sphingosine bond in C-16-ceramide results in approximately a 10-fold reduction in efficiency as a substrate for rat brain ceramidase compared to the unsaturated substrate. [4] However, experimental characterization of C12 NBD dihydro ceramide as a ceramidase substrate needs to be performed.
Reference:
[1]. Tani, M., Okino, N., Mori, K., et al. Molecular cloning of the full-length cDNA encoding mouse neutral ceramidase. A novel but highly conserved gene family of neutral/alkaline ceramidases. The Journal of Biological Chemisty 275(15), 11229-11234 (2000).
[2]. Ferlinz, K., Kopal, G., Bernardo, K., et al. Human acid ceramidase. Processing, glycosylation, and lysosomal targeting. The Journal of Biological Chemisty 276(38), 35352-35360 (2001).
[3]. Tani, M., Okino, N., Mitsutake, S., et al. Specific and sensitive assay for alkaline and neutral ceramidases involving C12-NBD-ceramide. J. Biochem. 125(4), 746-749 (1999).
[4]. Bawab, S.E., Usta, J., Roddy, P., et al. Substrate specificty of rat brain ceramidase. Journal of Lipid Research 43, 141-148 (2002).
Average Rating: 5 (Based on Reviews and 22 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















