Cilengitide (Synonyms: EMD 121974) |
| Catalog No.GC13559 |
Integrin inhibitor for αvβ3 and αvβ5
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 188968-51-6
Sample solution is provided at 25 µL, 10mM.
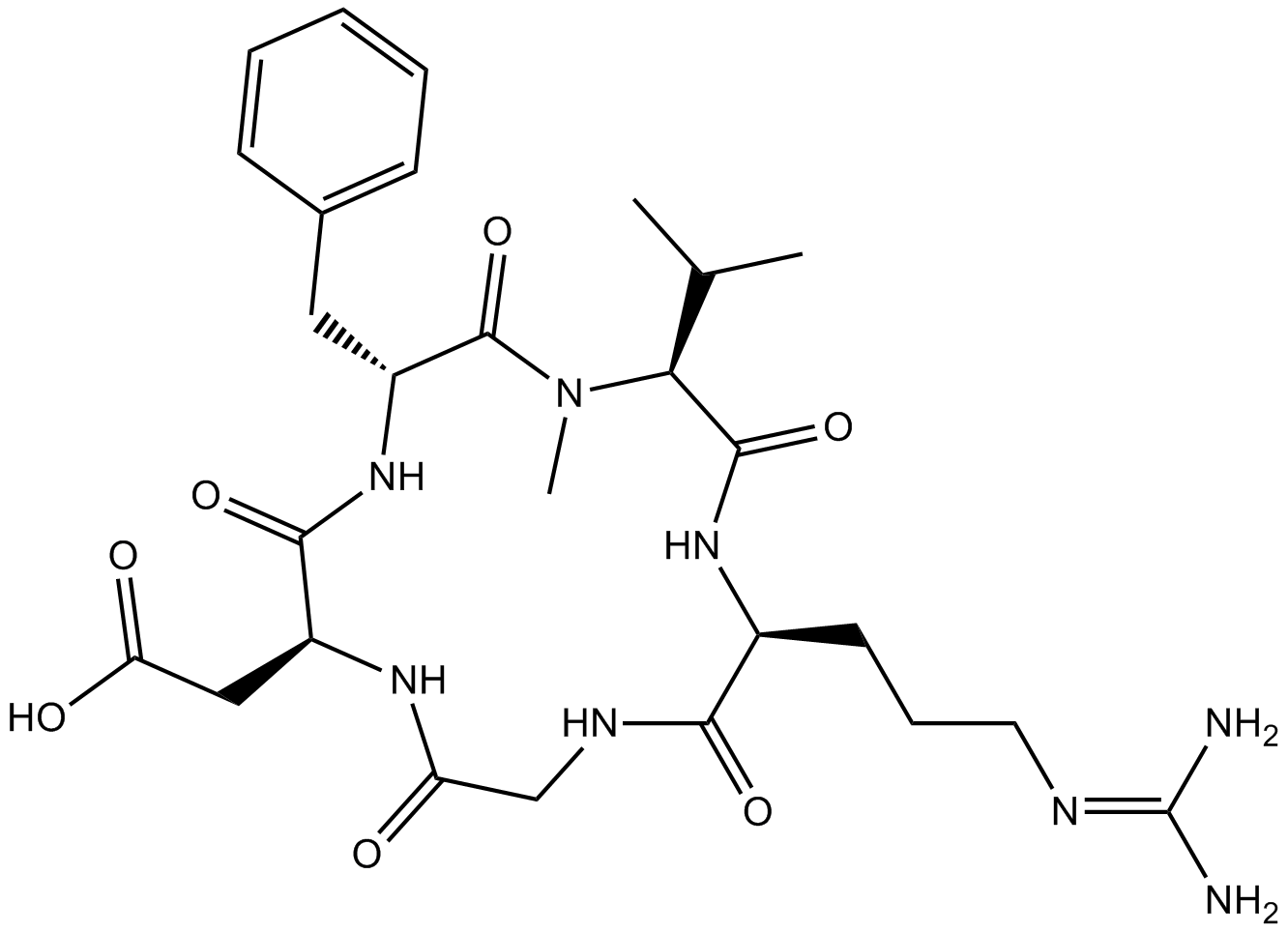
Cilengitide is a cyclic RGD pentapeptide [Arg-Gly-Asp-DPhe-(NMeVal)], and a potent αvβ3 and αvβ5 integrin inhibitor to block integrin-mediated adhesion and migration. It can directly bind αvβ3 integrin. Its IC50 is 250 nM as an αvβ3 inhibitor [1] [2] [3] [4].
Integrins are named for a family including 24 transmembrane heterodimer receptors that are composed of paired alpha and beta chains. These receptors can integrate extracellular and intracellular activities. Consequently, they can regulate tumor angiogenesis, migration and invasion [2].
When treated with cilengitide, a significant dose-dependent reduction of proliferation was noted (p<0.0002) in cell lines developed through 28 d of expansion and hence 14 d of differentiation culture of CD133+ stem cells (with VEGF, SCGF and FLT3L, to prepare CD133+ EPCs, i.e. endothelial progenitor cells). When treated with cilengitide after withdrawal of FLT3L and SCGF, a dose-dependent decrease of adherent cells was noted in EPCs after 7 and 14 days (p<0.03). Compared with which on EPC proliferation, the inhibitory effect of cilengitide on endothelial cell attachment was more pronounced [3].
Therapy with cilengitide intraperitoneally 5 times per week between days 1 and 30 after injection of MDA-MB-231 cells (105), volumes of osteolytic lesions(OL) and soft tissue components(SC) were significantly reduced on days 30 and 35 in rats, compared with untreated nude rats (p<0.05) [5].
References:
[1]. Carlos Mas-Moruno, Florian Rechenmacher and Horst Kessler. Cilengitide: The First Anti-Angiogenic Small Molecule Drug Candidate. Design, Synthesis and Clinical Evaluation. Anti-Cancer Agents in Medicinal Chemistry, 2010, 10(10): 753-768.
[2]. David A Reardon, L Burt Nabors, Roger Stupp, et al. Cilengitide: an integrin-targeting arginine-glycine-aspartic acid peptide with promising activity for glioblastoma multiforme. Expert Opin Investig Drugs, 2008, 17(8):1225-1235.
[3]. Sonja Loges, Martin Butzal, Jasmin Otten, et al. Cilengitide inhibits proliferation and differentiation of human endothelial progenitor cells in vitro. Biochemical and Biophysical Research Communications, 2007, 357: 1016-1020.
[4]. Despoina Sykoutri, Nisha Geetha, Silvia Hayer, et al. αvβ3 Integrin Inhibition with Cilengitide both Prevents and Treats Collagen Induced Arthritis. Ann Rheum Dis, 2013, 72(Suppl 1):A1-A88.
[5]. Maren Bretschi, Maximilian Merz, Dorde Komljenovic, et al. Cilengitide inhibits metastatic bone colonization in a nude rat model. Oncology Reports, 2011, 26:843-851.
Average Rating: 5 (Based on Reviews and 1 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















