(S)-Higenamine hydrobromide |
| Katalog-Nr.GC62751 |
(S)-Higenamin ((S)-Norcolaurin)-Hydrobromid, ein S-Enantiomer von Higenamin, ist die Einstiegsverbindung in die Benzylisochinolin-Alkaloid-Biosynthese.
Products are for research use only. Not for human use. We do not sell to patients.
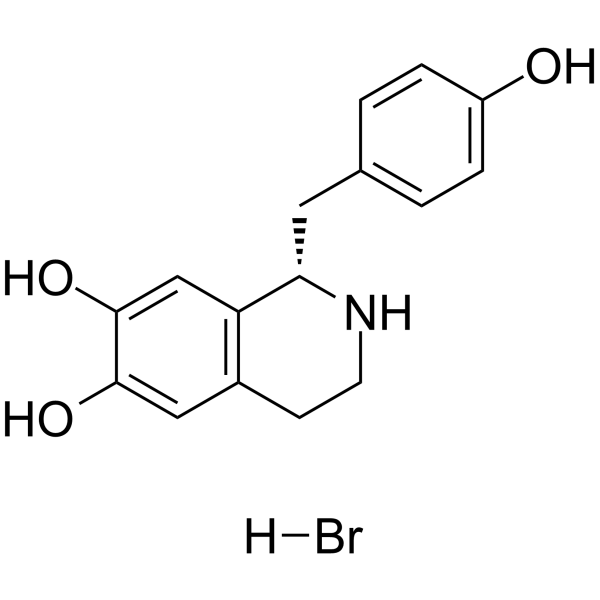
Cas No.: 105990-27-0
Sample solution is provided at 25 µL, 10mM.
(S)-Higenamine ((S)-Norcoclaurine) hydrobromide, a S-enantiomer of Higenamine, is the entry compound in benzylisoquinoline alkaloid biosynthesis. (S)-Higenamine hydrobromide is produced by the condensation of dopamine and 4-hydroxyphenylacetaldehyde (4-HPAA) by norcoclaurine synthase (NCS)[1].
The biosynthetic pathway leading to benzylisoquinoline alkaloids originates from the enzyme-catalyzed condensation of dopamine and 4-hydrophenylacetaldehyde to yield (S)-norcoclaurine. Both substrates are secondary metabolites derived from the decarboxylation/hydroxylation/deamination of tyrosine[1].
[1]. Minami H, et al. Functional analysis of norcoclaurine synthase in Coptis japonica. J Biol Chem. 2007;282(9):6274‐6282.
Average Rating: 5 (Based on Reviews and 17 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















