Efavirenz (Synonyms: DMP 266, EFV, L-743,726) |
| Catalog No.GC12389 |
Reverse transcriptase inhibitor
Products are for research use only. Not for human use. We do not sell to patients.
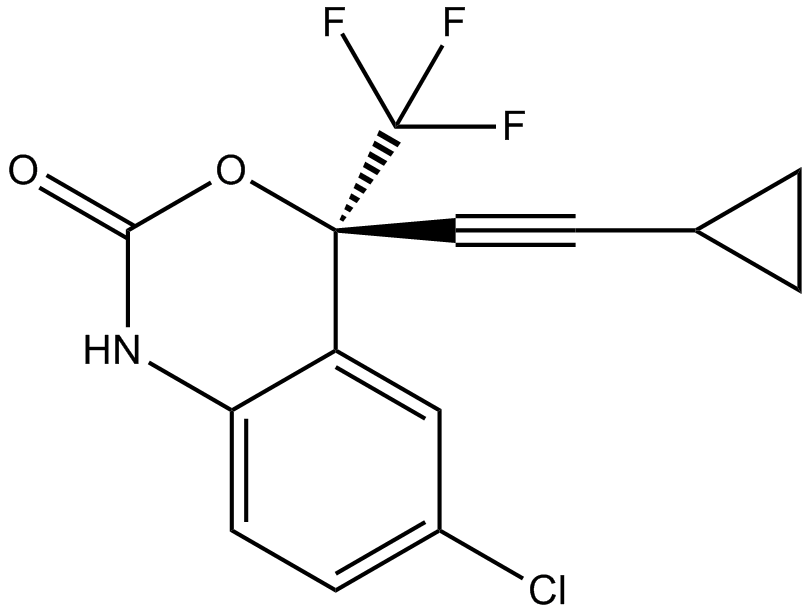
Cas No.: 154598-52-4
Sample solution is provided at 25 µL, 10mM.
Efavirenz is a highly potent inhibitor of human immunodeficiency virus type 1 reverse transcriptase with Ki value of 2.93nM [1].
Efavirenz is an antiretroviral drug approved by the FDA in 1998. It is usually used in a combination therapy with other antiretroviral drugs. Efavirenz is a potent inhibitor of both wild-type HIV-1 RT and HIV-1 variants which express series of NNRTI resistance-associated amino acid substitutions. The Ki value of efavirenz against the purified wild-type HIV-1 RT is 2.93nM. For the mutants A98G, L100I, K101G and K103N, the Ki values are 3.85nM, 17.13nM, 7.27nM and 17.6nM, respectively. Efavirenz is also a selective inhibitor of HIV-1 RT. It shows no inhibitory activity against a variety of polymerase enzymes including avian myeloblastosis virus RT, Moloney murine leukemia virus RT, human DNA polymerases and Escherichia coli RNA polymerase. In the acute infection assay, efavirenz also exerts potency with both wild-type HIV-1 RT (IC95s ranging from 1.5nM-3nM) and mutant HIV-1 RT [1, 2].
References:
[1] Young S D, Britcher S F, Tran L O, et al. L-743, 726 (DMP-266): a novel, highly potent nonnucleoside inhibitor of the human immunodeficiency virus type 1 reverse transcriptase. Antimicrobial agents and chemotherapy, 1995, 39(12): 2602-2605.
[2] de Béthune M P. Non-nucleoside reverse transcriptase inhibitors (NNRTIs), their discovery, development, and use in the treatment of HIV-1 infection: a review of the last 20 years (1989–2009). Antiviral research, 2010, 85(1): 75-90.
Average Rating: 5 (Based on Reviews and 20 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















