Dorsomorphin (Compound C) (Synonyms: Compound C) |
| Catalog No.GC17243 |
La dorsomorphine (Composé C) (Composé C) est un inhibiteur sélectif et compétitif de l'AMPK (Ki = 109 nM en l'absence d'AMP). La dorsomorphine (composé C) (BML-275) inhibe sélectivement les récepteurs BMP de type I ALK2, ALK3 et ALK6. La dorsomorphine (Composé C) induit l'autophagie.
Products are for research use only. Not for human use. We do not sell to patients.
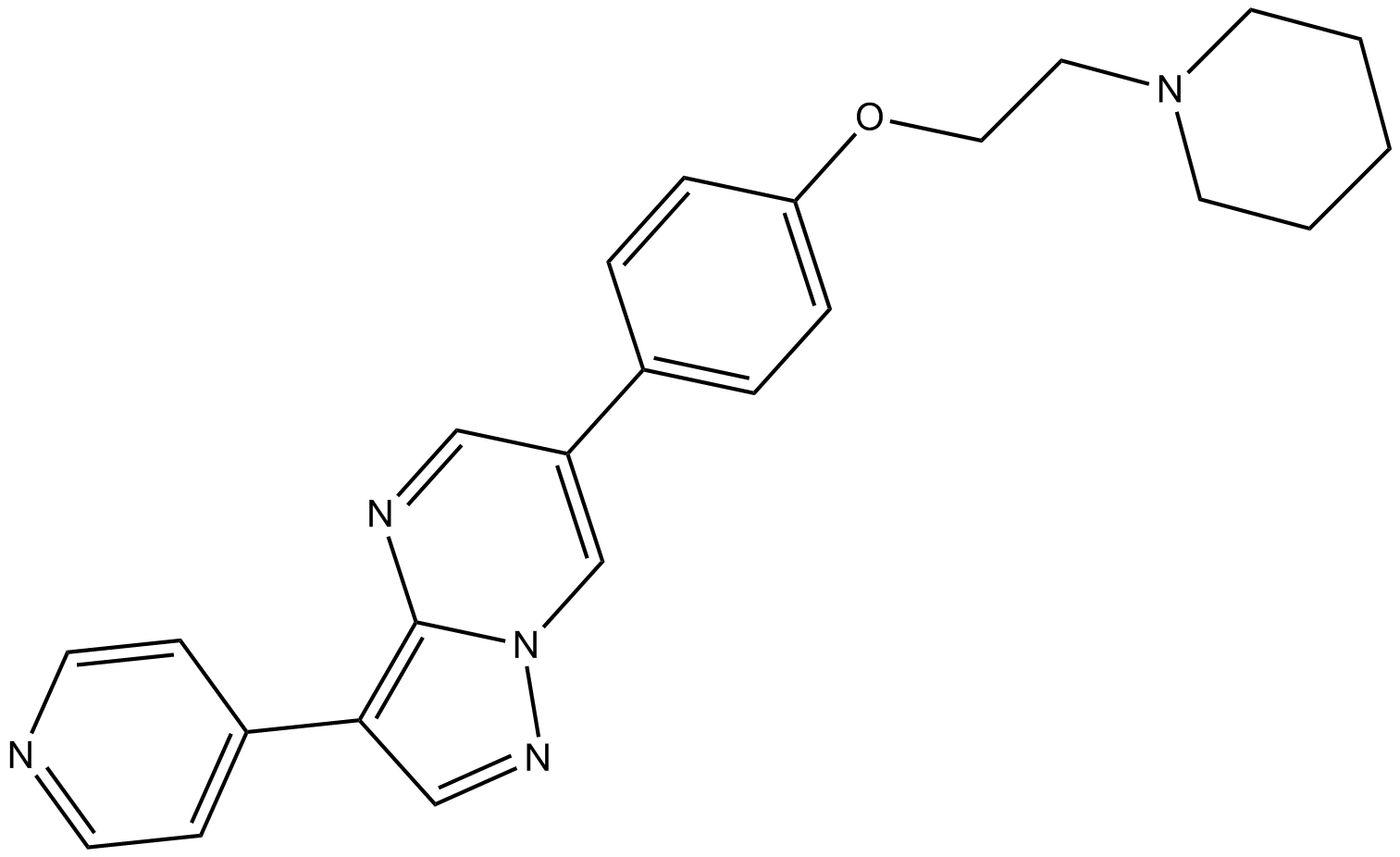
Cas No.: 866405-64-3
Sample solution is provided at 25 µL, 10mM.
Dorsomorphine (Composé C) est un agent utilisé comme inhibiteur de l'AMPK perméable à la cellule. Il pourrait sauver les actions antiprolifératives d'AICAR et de metformine. De plus, dorsomorphin (composé C) est également utilisé comme inhibiteur sélectif de la voie BMP. Le composé C pourrait inhiber un certain nombre de kinases autres que l'AMPK.[1]
Des expériences in vitro indiquent que le composé C inhibe l'activité de l'AMPK et la prolifération des cellules de gliome humain. Le dorsomorphine (composé C) réduit également l'apoptose des cellules induite par le cisplatine, et diminue l'expression de c-caspase3 et c-PARP lors du traitement au cisplatine.
Une étude in vivo démontre que le composé C atténue la néphrotoxicité induite par la cisplatine chez les souris et soulage l'activation de c-caspase 3 et c-PARP induite par la cisplatine dans les tissus rénaux. [1] [2]
References:
[1].Liu X, et al. The AMPK inhibitor compound C is a potent AMPK-independent antiglioma agent. Mol Cancer Ther. 2014 Mar;13(3):596-605.
[2].Li F, et al. Compound C Protects Against Cisplatin-Induced Nephrotoxicity Through Pleiotropic Effects. Front Physiol. 2020 Dec 23;11:614244.
Average Rating: 5 (Based on Reviews and 11 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




