Doxorubicin (Synonyms: Hydroxydaunorubicin) |
| Catalog No.GC16994 |
Doxorubicin(DOX), also known as adriamycin, is a compound of the anthracycline class that has the broadest spectrum of activity.
Products are for research use only. Not for human use. We do not sell to patients.
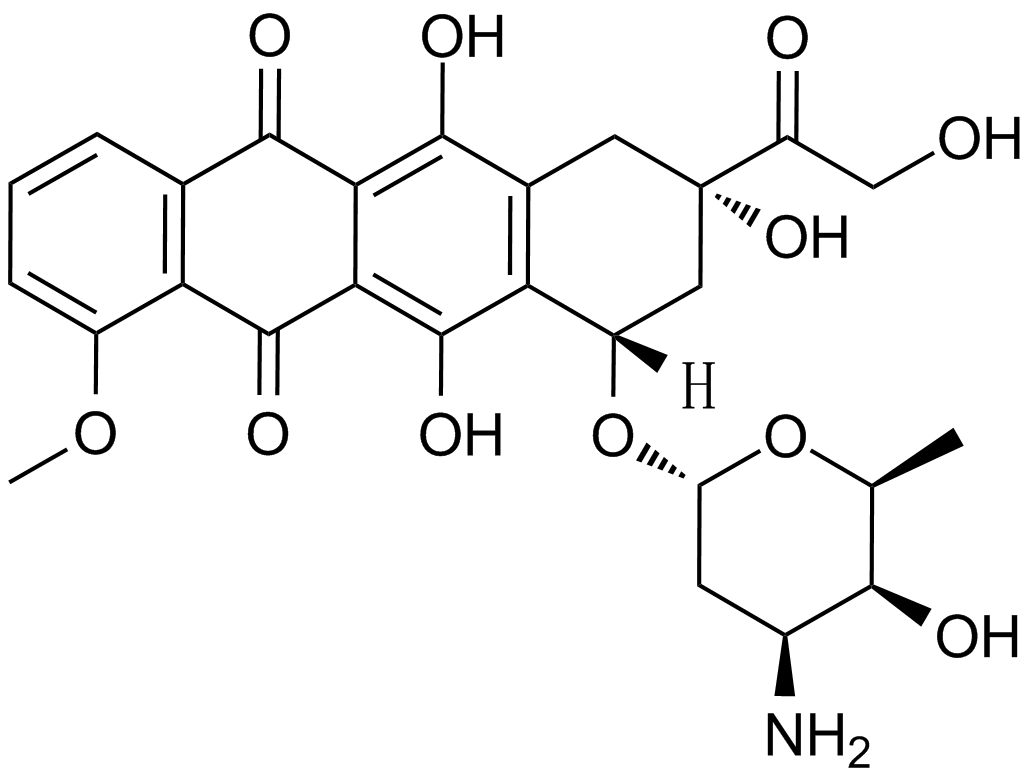
Cas No.: 23214-92-8
Sample solution is provided at 25 µL, 10mM.
Doxorubicin(DOX), also known as adriamycin, is a compound of the anthracycline class that has the broadest spectrum of activity[1]. Doxorubicin inhibits topoisomerase Ⅱ and topoisomerase Ⅰ with IC50 of 2.67μM and 0.8μM, respectively[2,3]. It is widely used for the treatment of various solid tumors via interacting with deoxyribonucleic acid, but it is limited in the clinical application due to severe side effect[4]
Doxorubicin can be loaded into liposomes by transmembrane pH gradient method to get high encapsulation efficiency with high drug/lipid ratio. Liposomal doxorubicin is a successful clinical formulation, and also a perfect model drug system for cancer-therapy research[5]. A considerable amount of doxorubicin can accumulate in human placental tissue. Both doxorubicin and its pH-sensitive liposomal formulation, L-Doxorubicin, are efficiently internalized by human trophoblastic BeWo cells and that doxorubicin accumulates in placental tissue so that decrease the exposure of fetal[6]
Doxorubicin combines with cobimetinib at sublethal dose completely arrested osteosarcoma growth. Targeted MEK inhibition by cobimetinib enhances doxorubicin’s efficacy in osteosarcoma models[7]. Doxorubicin is frequently used as an adjuvant chemotherapeutic agent for breast cancer. Silk films loaded with doxorubicin provide locoregional control of human breast cancer in vivo. By manipulating silk crystallinity or β-sheet content, the doxorubicin release rate could be controlled. Both soluble and stabilised silk films loaded with doxorubicin had a significantly greater primary tumour response than the equivalent dose of doxorubicin administered intravenously in the absence of the silk film carrier. The future use of this approach for localised chemotherapy is promising[8]
References:
[1].Escoffre JM, Piron J, et al. Doxorubicin delivery into tumor cells with ultrasound and microbubbles. Mol Pharm. 2011;8(3):799-806.
[2].Rhee HK, Park HJ, et al. Synthesis, cytotoxicity, and DNA topoisomerase II inhibitory activity of benzofuroquinolinediones. Bioorg Med Chem. 2007 Feb 15;15(4):1651-8.
[3].Foglesong PD, Reckord C, Swink S. Doxorubicin inhibits human DNA topoisomerase I. Cancer Chemother Pharmacol. 1992;30(2):123-5.
[4].Jie L, Lang D, et al. Superparamagnetic Iron Oxide Nanoparticles/Doxorubicin-Loaded Starch-Octanoic Micelles for Targeted Tumor Therapy. J Nanosci Nanotechnol. 2019;19(9):5456-5462.
[5].Soininen SK, Repo JK, et al. Human placental cell and tissue uptake of doxorubicin and its liposomal formulations. Toxicol Lett. 2015;239(2):108-114.
[6].Niu G, Cogburn B, et al. Preparation and characterization of doxorubicin liposomes. Methods Mol Biol. 2010;624:211-219.
[7].Seib FP, Kaplan DL. Doxorubicin-loaded silk films: drug-silk interactions and in vivo performance in human orthotopic breast cancer. Biomaterials. 2012;33(33):8442-8450.
[8]. Ma L, Xu Y, Xu X. Targeted MEK inhibition by cobimetinib enhances doxorubicin's efficacy in osteosarcoma models. Biochem Biophys Res Commun. 2020;529(3):622-628.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




