GSK2239633A |
| Catalog No.GC31744 |
GSK2239633A est un antagoniste du récepteur CC-chimiokine 4 (CCR4), qui inhibe la liaison du [125I]-TARC au CCR4 humain avec une pIC50 de 7,96±0,11.
Products are for research use only. Not for human use. We do not sell to patients.
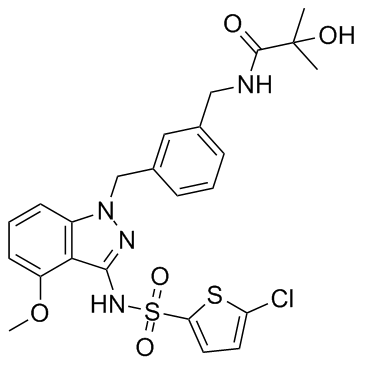
Cas No.: 1240516-71-5
Sample solution is provided at 25 µL, 10mM.
GSK2239633A is a CC-chemokine receptor 4 (CCR4) antagonist, which inhibits the binding of [125I]-TARC to human CCR4 with a pIC50 of 7.96 ± 0.11.
The GSK2239633A is an allosteric antagonist of human CCR4. GSK2239633A inhibits the binding of [125I]-TARC to human CCR4 with a pIC50 of 7.96±0.11 and also inhibits thymus- and activation-regulated chemokine-induced (TARC)-induced increases in the F-actin content of isolated human CD4+ CCR4+ T-cells with a pA2 of 7.11±0.29[1]. The effect of GSK2239633A (Compound 3) on CCL17-induced increases in the F-actin content of human CD4+ CCR4+ T cells is measured. The pEC50 value is 8.79±0.22[2].
Following intravenous dosing, plasma GSK2239633A displays rapid, bi-phasic distribution and slow terminal elimination (t1/2: 13.5 hours), suggesting that GSK2239633A is a low to moderate clearance drug. Following oral dosing, blood levels of GSK2239633A reach Cmax rapidly (median tmax: 1.0-1.5 hours). Estimated GSK2239633A bioavailability is low with a maximum value determined of only 16%[1]. GSK2239633A (Compound 9) demonstrates good pharmacokinetic data in preclinical animal studies (bioavailability in rats and beagle dogs 85% and 97% respectively)[3].
[1]. Cahn A, et al. Safety, tolerability, pharmacokinetics and pharmacodynamics of GSK2239633, a CC-chemokine receptor 4 antagonist, in healthy male subjects: results from an open-label and from a randomised study. BMC Pharmacol Toxicol. 2013 Feb 28;14:14. [2]. Slack RJ, et al. Antagonism of human CC-chemokine receptor 4 can be achieved through three distinct binding sites on the receptor. Pharmacol Res Perspect. 2013 Dec;1(2):e00019. [3]. Miah AH, et al. Identification of pyrazolopyrimidine arylsulfonamides as CC-chemokine receptor 4 (CCR4) antagonists. Bioorg Med Chem. 2017 Oct 15;25(20):5327-5340.
Average Rating: 5 (Based on Reviews and 24 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















