Fumarate hydratase-IN-1 |
| Catalog No.GC32878 |
Fumarate hydratase-IN-1 (compound 2) is a cell-permeable fumarate hydratase inhibitor. Fumarate hydratase-IN-1 has antiproliferative activity against several cancer cell lines with a mean IC50 of 2.2 μM.
Products are for research use only. Not for human use. We do not sell to patients.
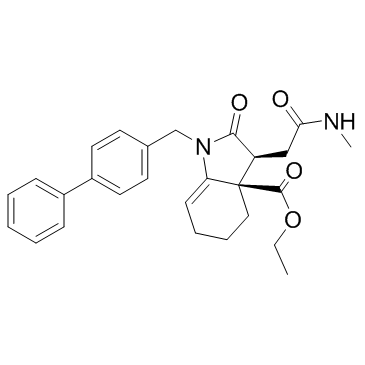
Cas No.: 1644060-37-6
Sample solution is provided at 25 µL, 10mM.
Fumarate hydratase-IN-1, an enzyme of the TCA cycle. Inhibition of fumarate hydratase-IN-1 can contribute to tumorigenicity in some cells. The use of a photoaffinity labeling strategy identified fumarate hydratase as the principal pharmacological target.[1]In vitro: The activity of this enzyme was measured using a well-established assay that monitored the conversion of fumarate into L-malate and subsequent oxidation of L-malate to oxaloacetate by malate dehydrogenase. Initial controls established that neither the carboxylic acid 3 nor ester 2 inhibited malate dehydrogenase Using this two-enzyme protocol we found that carboxylic acid 3 inhibited fumarate hydratase in a dose-dependent fashion in vitro. Besides, inhibition of fumarate hydratase can contribute to tumorigenicity in some cells.[1]
[1]. Takeuchi T et al. Identification of Fumarate Hydratase Inhibitors with Nutrient-Dependent Cytotoxicity. J Am Chem Soc, 2015 Jan 21, 137(2): 564-567.
Average Rating: 5 (Based on Reviews and 13 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















