Gamitrinib TPP hexafluorophosphate |
| Catalog No.GC32766 |
Gamitrinib TPP hexafluorophosphate is a Gamitrinib (GA) mitochondrial matrix inhibitor. Gamitrinib TPP hexafluorophosphate is a mitochondrial targeted HSP90 inhibitor with anti-cancer activity.
Products are for research use only. Not for human use. We do not sell to patients.
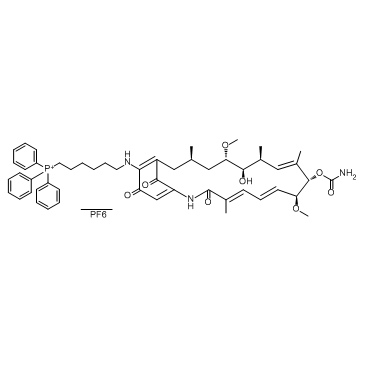
Cas No.: 1131626-47-5
Sample solution is provided at 25 µL, 10mM.
Gamitrinib TPP hexafluorophosphate is a Gamitrinib (GA) mitochondrial matrix inhibitor.
Gamitrinib TPP (GamitrinibTPP, G-TPP), a small molecule that combines the Hsp90 ATPase inhibitory module of 17-allylamino geldanamycin (17-AAG) with the mitochondrial-targeting moiety of triphenylphosphonium. Gamitrinib TPP is selectively delivered to mitochondria and does not affect Hsp90 homeostasis outside the organelle. Within a 16-hour exposure, concentrations of Gamitrinib TPP of 15-20 μM indistinguishably kill patient-derived and cultured glioblastoma cell lines. This cell death response has the hallmarks of mitochondrial apoptosis, with loss of organelle inner membrane potential, release of cytochrome c in the cytosol, activation of initiator caspase-9 and effector caspase-3 and caspase-7, and cellular reactivity for annexin V[1].
Whether the combination of TRAIL plus Gamitrinib TPP (GamitrinibTPP, G-TPP) has activity against glioblastoma in vivo is studied. Luciferase-expressing U87 glioblastoma cells implanted in the right cerebral striatum of immunocompromised mice give rise to rapidly growing tumors by bioluminescence imaging, and treatment of these mice with vehicle, stereotactic delivery of TRAIL, or systemic administration of suboptimal concentrations of Gamitrinib TPP does not affect tumor growth in vivo. Similarly, systemic monotherapy with Gamitrinib TPP at concentrations (20 mg/kg as daily i.p. injections) that inhibit subcutaneous xenograft tumor growth in mice has no effect on orthotopic glioblastoma growth. In contrast, 2 cycles of intracranial TRAIL combined with systemic Gamitrinib TPP suppresses the growth of established glioblastomas, with no significant animal weight loss throughout treatment[1].
[1]. Markus D. Siegelin, et al. Exploiting the mitochondrial unfolded protein response for cancer therapy in mice and human cells. J Clin Invest. 2011 Apr 1; 121(4): 1349-1360.
Average Rating: 5 (Based on Reviews and 6 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















