GW3965 HCl |
| Catalog No.GC17696 |
GW3965 HCl is a potent and selective liver X receptor (LXR) agonist with EC50s of 190 nM and 30 nM for hLXRα and hLXRβ, respectively.
Products are for research use only. Not for human use. We do not sell to patients.
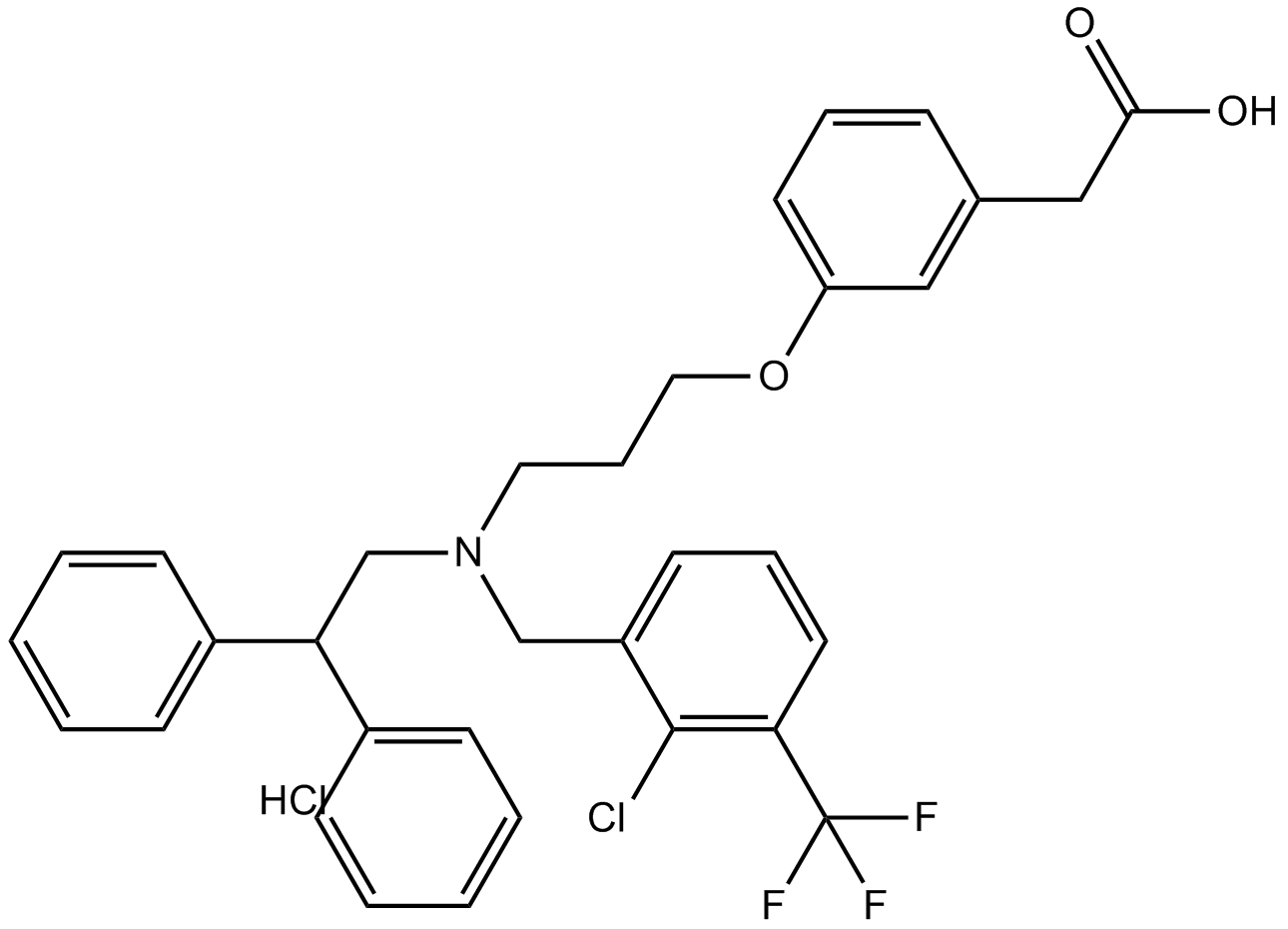
Cas No.: 405911-17-3
Sample solution is provided at 25 µL, 10mM.
GW3965 HCl is a selective, orally active non-steroidal agonist for the liver X receptor (LXR) [1].
Nuclear receptors LXRα and LXRβ have an important role in control of cholesterol and fatty acid metabolism in multiple cell types. Activation of these receptors promotes bile acid synthesis in liver, inhibits intestinal cholesterol absorption and stimulates cholesterol efflux in macrophages, which will then reduce atherosclerotic risk [2].
In a cell-free ligand-sensing assay, GW3965 recruits the steroid receptor coactivator 1 to human LXRR with an EC50 of 125 nM. In cell-based reporter gene assays, GW3965 plays as a full agonist on hLXRα and hLXRβ with EC50 of 190 and 30 nM, respectively [1].
Treatment C57BL/6 mice with 10 mg/kg GW3965 HCl orally, GW3965 HCl increased the plasma concentrations of HDL cholesterol by 30% and enhanced the expression of the reverse cholesterol transporter ABCA1 in the small intestine and peripheral macrophages [1]. In male Sprague–Dawley rats, GW3965 decreased Ang II-mediated vasopressor responses and reduced ATR gene expression, which suggested GW3965 can affect vascular reactivity [3].
References:
[1]. Collins JL, Fivush AM, Watson MA, et al. Identification of a nonsteroidal liver X receptor agonist through parallel array synthesis of tertiary amines. J Med Chem, 2002, 45(10): 1963-1966.
[2]. Joseph SB, McKilligin E, Pei L, et al. Synthetic LXR ligand inhibits the development of atherosclerosis in mice. Proc Natl Acad Sci U S A, 2002, 99(11): 7604-7609.
[3]. Leik CE, Carson NL, Hennan JK, et al. GW3965, a synthetic liver X receptor (LXR) agonist, reduces angiotensin II-mediated pressor responses in Sprague-Dawley rats. Br J Pharmacol, 2007, 151(4): 450-456.
Average Rating: 5 (Based on Reviews and 33 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















