JLK 6 (Synonyms: JLK 6) |
| Catalog No.GC13619 |
A protease inhibitor
Products are for research use only. Not for human use. We do not sell to patients.
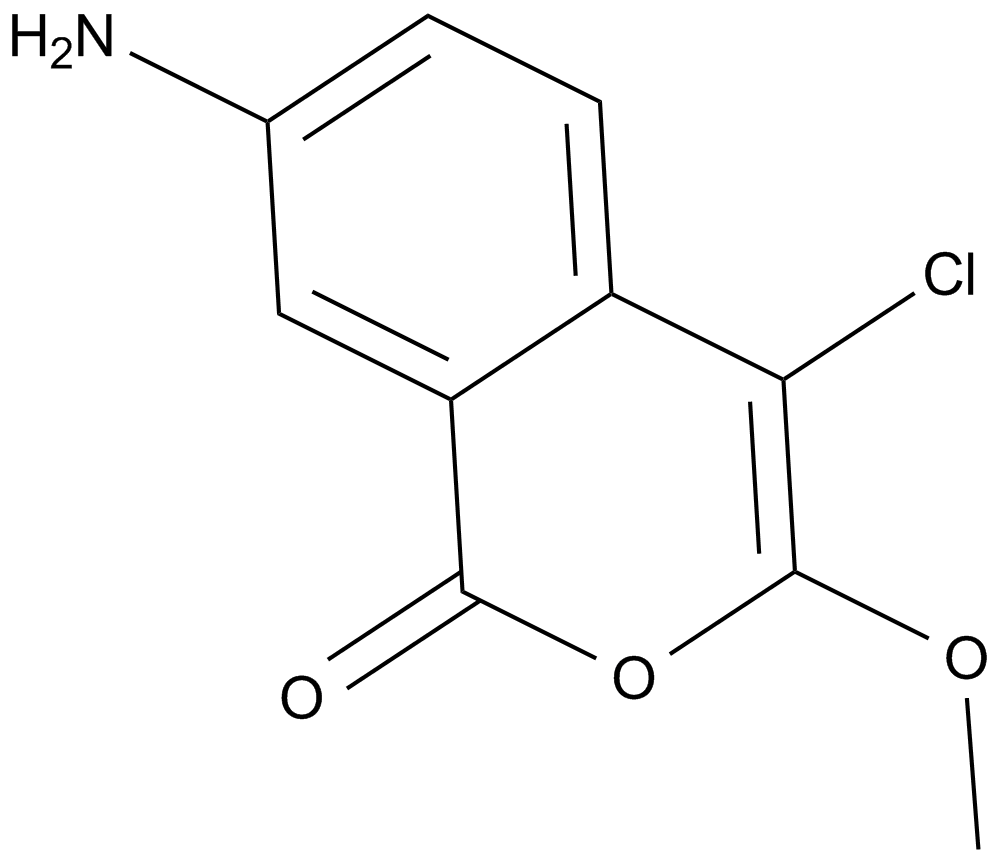
Cas No.: 62252-26-0
Sample solution is provided at 25 µL, 10mM.
JLK 6, an isocoumarin, is a selective inhibitor of γ-secretase, with an IC50 value between 10 μM-1 mM [1, 2].
The enzyme γ-secretase catalyzes the cleavage of β-Amyloid precursor protein (βAPP) to produce Amyloid β-peptide (Aβ). Aβ is a part of the plaque present in the brain of patients with Alzheimer’s disease. γ-secretase also targets other substrates like Notch. Notch is a transmembrane protein which is involved in important functions during different stages in development, both embryonic and adulthood [1].
HEK293 cells were used. In these cells, wild-type βAPP was overexpressed (962 fmol/mL in 35-mm wells). JLK6 markedly reduced Aβ secreted from these cells. Interestingly, JLK6 potentiated the recovery of two fragments. Immunological characterization indicated that one fragment was labelled with a specific antibody against the Asp1 residue of Aβ. JLK6 also inhibited the Aβ recovery from cells overexpressing Swedish-mutant βAPP to a similar extent [2].
In the zebrafish embryo, JLK isocoumarin inhibitors did not change the Notch pathway responsible for somitogenesis. Unlike other γ-secretase inhibitors, these agents did not affect E-cadherin processing. JLKs did not inhibit α-secretase, β-site APP cleaving enzymes (BACE) 1 and BACE2, GSK3β kinase and proteasome. JLK inhibitors prevented Aβ production without inducing unwanted cleavages of other proteins [1].
References:
[1]. Petit A, Pasini A, Alves Da Costa C, et al. JLK isocoumarin inhibitors: Selective γ-secretase inhibitors that do not interfere with notch pathway in vitro or in vivo. Journal of neuroscience research, 2003, 74(3): 370-377.
[2]. Petit A, Bihel F, da Costa CA, et al. New protease inhibitors prevent γ-secretase-mediated production of Aβ40/42 without affecting Notch cleavage. Nature cell biology, 2001, 3(5): 507-511.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















