L-Eflornithine (L-DFMO) |
| Catalog No.GC33414 |
L-Eflornithine (L-DFMO) (L-DFMO) is an enantiomer of Eflornithine. L-Eflornithine (L-DFMO) is an irreversible ornithine decarboxylase (ODC) inhibitor with a KD of 1.3±0.3 μM, and a Kinact of 0.15±0.03 min-1.
Products are for research use only. Not for human use. We do not sell to patients.
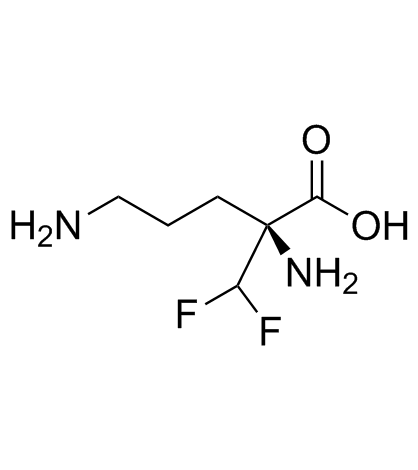
Cas No.: 66640-93-5
Sample solution is provided at 25 µL, 10mM.
L-Eflornithine (L-DFMO) is an enantiomer of Eflornithine. L-Eflornithine is an irreversible ornithine decarboxylase (ODC) inhibitor with a KD of 1.3±0.3 µM, and a Kinact of 0.15±0.03 min-1[1].
Eflornithine (D/L-DFMO) is an inhibitor of ODC, the first enzyme in eukaryotic polyamine biosynthesis. Both enantiomers of Eflornithine (DFMO) irreversibly inactivate ODC. Both Eflornithine enantiomers (L-Eflornithine and D-Eflornithine) suppress ODC activity in a time- and concentration-dependent manner. The inhibitor dissociation constant (KD) values for the formation of enzyme-inhibitor complexes are 28.3±3.4, 1.3±0.3 and 2.2±0.4 µM respectively for D-Eflornithine, L-Eflornithine and Eflornithine. The inhibitor inactivation constants (Kinact) for the irreversible step were 0.25±0.03, 0.15±0.03 and 0.15±0.03 min-1 respectively for D-Eflornithine, L-Eflornithine and Eflornithine. Treatment of human colon tumour-derived HCT116 cells with either L-Eflornithine or D- Eflornithine decreases the cellular polyamine contents in a concentration-dependent manner[1]. The enantiomers display different potencies in vitro, with the L-enantiomer having up to a 20-fold higher affinity for the target enzyme ornithine decarboxylase[2]. The L-Eflornithine also appears to be more potent in cultured T.brucei gambiense parasites[2].
The more potent L-Eflornithine is present at much lower concentrations in both plasma and cerebrospinal fluid (CSF) than those of the D-Eflornithine. The plasma concentrations of L-Eflornithine are on average 52% of the D-enantiomer concentrations. The typical oral clearances of L-Eflornithine and D-eflornithine are 17.4 and 8.23 liters/h, respectively[2].
[1]. Qu N, et al. Inhibition of human ornithine decarboxylase activity by enantiomers of difluoromethylornithine. Biochem J. 2003 Oct 15;375(Pt 2):465-70. [2]. Jansson-L?fmark R, et al. Enantiospecific reassessment of the pharmacokinetics and pharmacodynamics of oral eflornithine against late-stage Trypanosoma brucei gambiense sleeping sickness. Antimicrob Agents Chemother. 2015 Feb;59(2):1299-307.
Average Rating: 5 (Based on Reviews and 14 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















