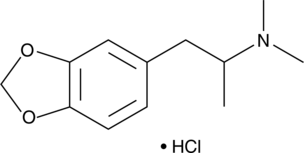
MDDMA (hydrochloride) (Synonyms: 3,4-Methylenedioxydimethylamphetamine) |
| Catalog No.GC44142 |
MDDMA is a centrally active substituted methylenedioxyphenethylamine that has appeared in illicit drug samples.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 74341-79-0
Sample solution is provided at 25 µL, 10mM.
MDDMA is a centrally active substituted methylenedioxyphenethylamine that has appeared in illicit drug samples. It is structurally similar to 3,4-MDMA with an additional methyl group that is predicted to result in relatively reduced central activity compared to 3,4-MDMA. This product is intended for research and forensic applications.
Average Rating: 5 (Based on Reviews and 13 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















