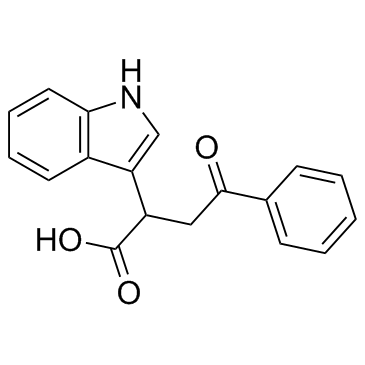
PEO-IAA (2-(1H-Indol-3-yl)-4-oxo-4-phenyl-butyric acid) |
| Catalog No.GC30168 |
PEO-IAA (2-(1H-Indol-3-yl)-4-oxo-4-phenyl-butyric acid) is an indole-3-acetic acid (IAA) antagonist.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 6266-66-6
Sample solution is provided at 25 µL, 10mM.
PEO-IAA is an indole-3-acetic acid (IAA) antagonist. PEO-IAA is an auxin antagonist that binds to transport inhibitor response 1/auxin signaling F-box proteins (TIR1/AFBs).
PEO-IAA is also an α-alkyl-IAA, and shows more potent anti-auxin activity in auxin-responsive gene expression and in the cell division and elongation pathway that is mediated via SCFTIR1/AFBs. PEO-IAA suppresses not only the expression of an auxin-responsive ZmSAUR2 gene, but also gravitropic curvature. PEO-IAA blocks the auxin response in Arabidopsis, rice, moss and maize[1].
[1]. Nishimura T, et al. Differential downward stream of auxin synthesized at the tip has a key role in gravitropic curvature via TIR1/AFBs-mediated auxin signaling pathways. Plant Cell Physiol. 2009 Nov;50(11):1874-85.
Average Rating: 5 (Based on Reviews and 34 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















