Toremifene Citrate (Synonyms: GTx-006, NK 622, NSC 613680) |
| Catalog No.GC13835 |
Oral selective estrogen receptor modulator (SERM)
Products are for research use only. Not for human use. We do not sell to patients.
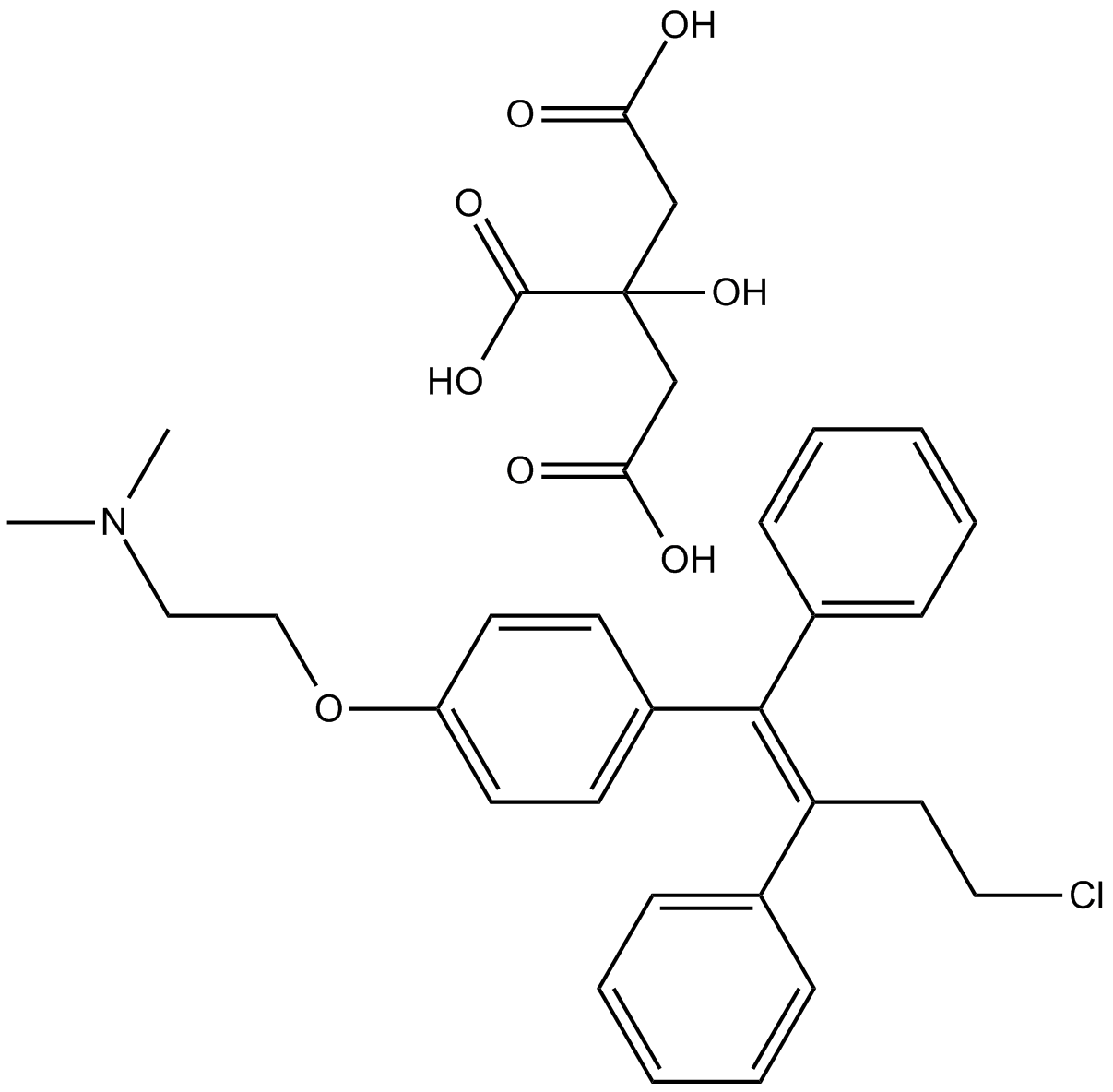
Cas No.: 89778-27-8
Sample solution is provided at 25 µL, 10mM.
Toremifene Citrate(NK 622; FC 1157a) is a second-generation selective estrogen-receptor modulator (SERM) in development for the prevention of osteoporosis.IC50 Value: 1±0.3 μMTarget: Estrogen receptorToremifene is a second-generation selective estrogen-receptor modulator (SERM) in development for the prevention of osteoporosis and other adverse effects resulting from ADT in men with prostate cancer [1]. in vitro: The growth of Ac-1 cells was inhibited by tamoxifen, toremifene and atamestane in vitro with IC50values of 1.8±1.3μM, 1±0.3μM and 60.4±17.2μM, respectively. The combination of toremifene plusatamestane was found to be better than toremifene or atamestane alone in vitro[2].in vivo: The effect of this combination was then studied in vivo using Ac-1 xenografts grown in ovariectomized female SCID mice. The mice were injected with toremifene (1000μg/day), atamestane (1000μg/day), tamoxifen (100μg/day), or the combination of toremifene plus atamestane. In this study, our results indicate that the combination of toremifene plus atamestane was as effective as toremifene or tamoxifen alone but may not provide any additional benefit over toremifene alone or tamoxifen alone[2].Clinical trail: Prostate cancer diagnosis among men with isolated high-grade intraepithelial neoplasia enrolled onto a 3-year prospective phase III clinical trial of oral toremifene[3].
References:
[1]. Matthew R Smith, Selective Estrogen Receptor Modulators to Prevent Treatment-Related Osteoporosis.Rev Urol. 2005; 7(Suppl 3): S30-S35.
[2]. Gauri J Sabnis, Luciana Macedo, Olga Goloubeva, Toremifene - Atamestane; Alone or In Combination: Predictions from the Preclinical Intratumoral Aromatase Model. J Steroid Biochem Mol Biol. 2008 January; 108(1-2): 1-7.
[3]. Taneja SS, Morton R, Barnette G, Prostate cancer diagnosis among men with isolated high-grade intraepithelial neoplasia enrolled onto a 3-year prospective phase III clinical trial of oral toremifene. J Clin Oncol. 2013 Feb 10;31(5):523-9.
Average Rating: 5 (Based on Reviews and 15 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















