Ziprasidone |
| Catalog No.GC11020 |
Products are for research use only. Not for human use. We do not sell to patients.
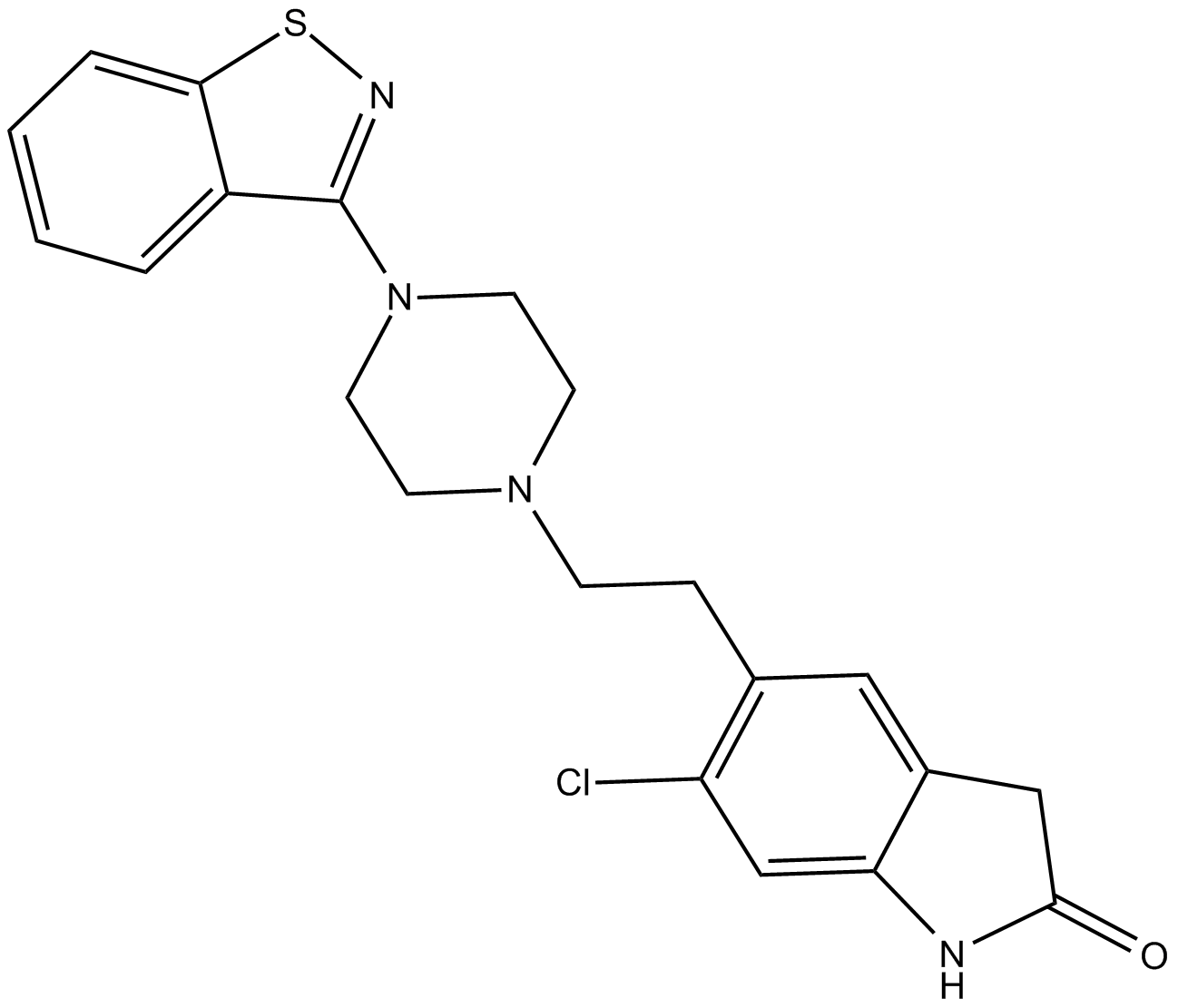
Cas No.: 146939-27-7
Sample solution is provided at 25 µL, 10mM.
Ziprasidone (CP-88059) is a combined 5-HT (serotonin) and dopamine receptor antagonist. Ziprasidone exhibits potent effects of antipsychotic activity, and is used for treating various mental disorders including schizophrenia[1][2][3].
Ziprasidone possesses an in vitro 5-HT2A/dopamine D2 receptor affinity ratio higher than any clinically available antipsychotic agent. In vivo, ziprasidone antagonizes 5-HT2A receptor-induced head twitch with 6-fold higher potency than for blockade of d-amphetamine-induced hyperactivity, a measure of central dopamine D2 receptor antagonism. Ziprasidone also has high affinity for the 5-HT1A, 5-HT1D and 5-HT2C receptor subtypes, which may further enhance its therapeutic potential[1]. Ziprasidone sulfoxide and sulfone were the major metabolites in human serum. The affinities of the sulfoxide and sulfone metabolites for 5-HT2 and D2 receptors are low with respect to ziprasidone, and are thus unlikely to contribute to its antipsychotic effects[2].Ziprasidone was associated with significant differential adverse effects relative to placebo in BPM, BPD, and schizophrenia with no significant difference in weight gain in all 3 groups. Self-reported somnolence was increased across the 3 conditions. Subjects with BPM were more vulnerable to EPS than those with BPD or schizophrenia[3].
References:
[1]. Seeger, T.F., et al., Ziprasidone (CP-88,059): a new antipsychotic with combined dopamine and serotonin receptor antagonist activity. J Pharmacol Exp Ther, 1995. 275(1): p. 101-13.
[2]. Prakash, C., et al., Metabolism and excretion of a new antipsychotic drug, ziprasidone, in humans. Drug Metab Dispos, 1997. 25(7): p. 863-72.
[3]. Gao, K., et al., Risk for adverse events and discontinuation due to adverse events of ziprasidone monotherapy relative to placebo in the acute treatment of bipolar depression, mania, and schizophrenia. J Clin Psychopharmacol, 2013. 33(3): p. 425-31.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




