Jasplakinolide (Synonyms: NSC 613009) |
| Catalog No.GC17312 |
stabilizes pre-formed actin filaments and inhibits their disassembly
Products are for research use only. Not for human use. We do not sell to patients.
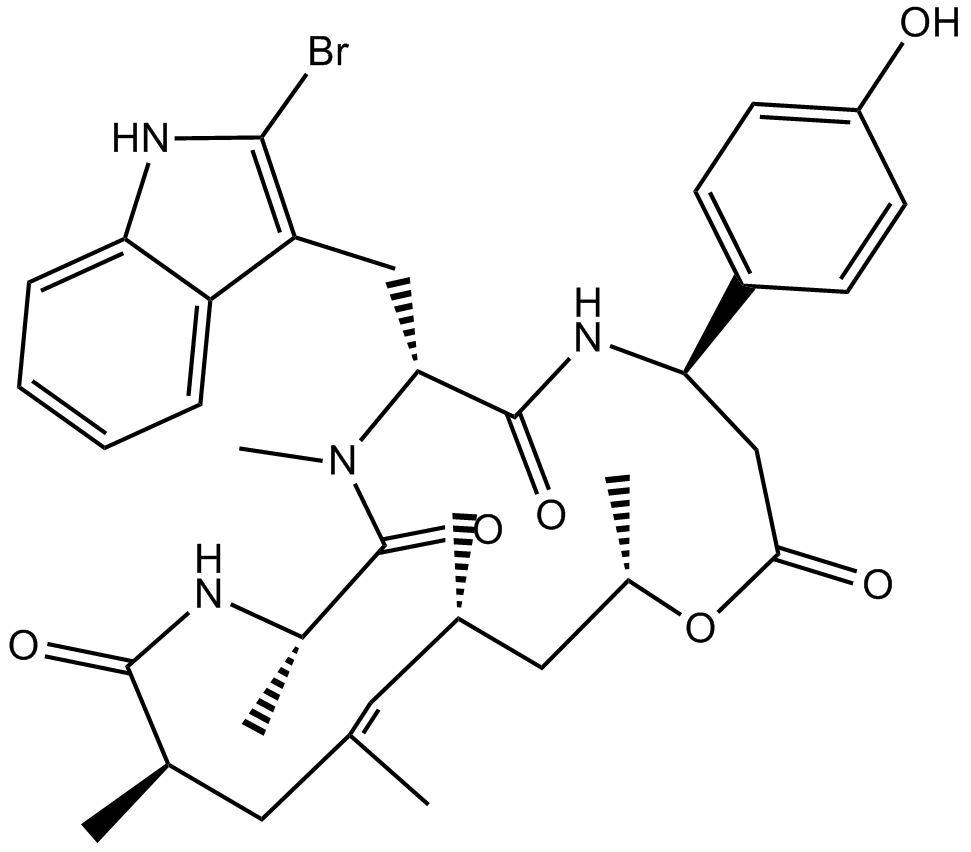
Cas No.: 102396-24-7
Sample solution is provided at 25 µL, 10mM.
Jasplakinolide is a natural macrocyclic peptide first isolated from a marine sponge. It potently inhibits the proliferation of PC3 prostate carcinoma cells (IC50 = 35 nM) by binding F-actin (KD = 15 nM).[1] This binding of jasplakinolide to actin, which is competitive with phalloidin, stabilizes actin filaments in vitro but disrupts actin filaments and induces irregular polymerization of monomeric actin in vivo.[1],[2] This compound is used to investigate the role of actin in diverse cellular roles, such as motility, transport, and development.[3],[4]
Reference:
[1]. Bubb, M.R., Senderowicz, A.M.J., Sausville, E.A., et al. Jasplakinolide, a cytotoxic natural product, induces actin polymerization and competitively inhibits the binding of phalloidin to F-actin. The Journal of Biological Chemisty 269(21), 14869-14871 (1994).
[2]. Bubb, M.R., Spector, I., Beyer, B.B., et al. Effects of jasplakinolide on the kinetics of actin polymerization. An explanation for certain in vivo observations. The Journal of Biological Chemisty 275(7), 5163-5170 (2000).
[3]. Van Goor, D., Hyland, C., Schaefer, A.W., et al. The role of actin turnover in retrograde actin network flow in neuronal growth cones. PLoS One 7(2), (2012).
[4]. Zhang, X., Cui, X., Cheng, L., et al. Actin stabilization by jasplakinolide affects the function of bone marrow-derived late endothelial progenitor cells. PLoS One 7(11), (2012).
Average Rating: 5 (Based on Reviews and 35 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















