HA-155 (Synonyms: Autotaxin Inhibitor IV) |
| Catalog No.GC10869 |
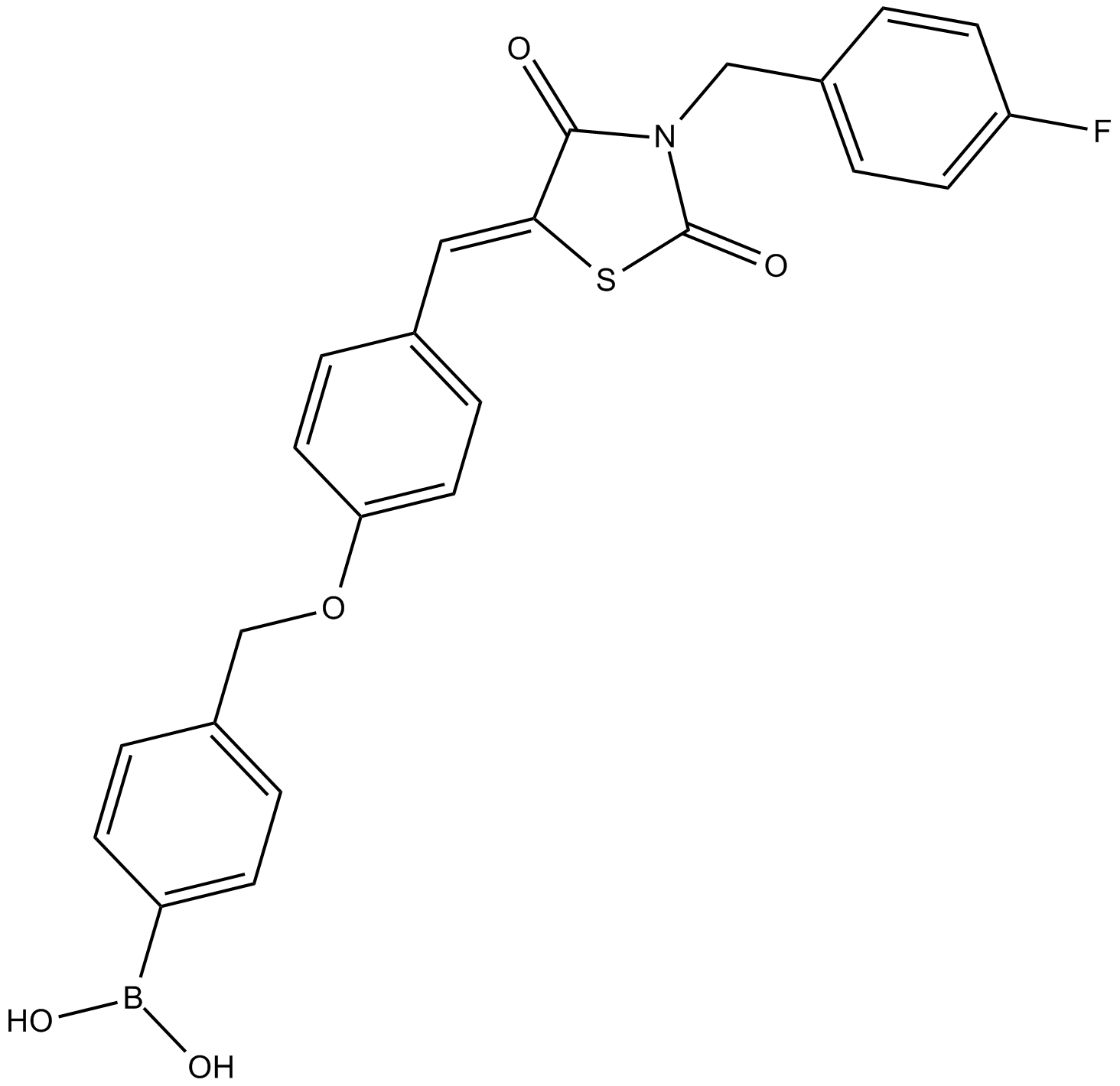
HA-155 is a potent autotaxin (ATX) type I inhibitor.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 1229652-22-5
Sample solution is provided at 25 µL, 10mM.
IC50 = 5.7 nM
HA155 is an autotaxin inhibitor.
Autotaxin (ATX), a secreted phosphodiesterase, hydrolyzes the abundant phospholipid lysophosphatidylcholine (LPC) to produce lysophosphatidic acid (LPA). The ATX-LPA signaling axis has been reported to be involved in fibrosis, inflammation, and tumor progression, rendering ATX an attractive drug target.
In vitro: HA155 was identified as a boronic acid-based inhibitor of ATX based on the crystal structure of ATX in complex with HA155. Furthermore, the syntheses and activities of HA155 could be explained by structural data. In order to further understand the difference in activity, molecular docking experiments were performed. The molecular docking indicated a remarkable binding pose for one of the isomers, which differed from the original binding pose of HA155 for ATX. Moreover, the thrombin-mediated increase in platelet-derived LPA was completely attenuated in a dose-dependent manner by HA155. HA-155 could inhibit autotaxin by selectively binding to its catalytic threonine. HA155 showed to dose-dependently block thrombin-induced LPA secretion in platelets [1,2].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Albers, H. M.H.G.,Hendrickx, L.J.D.,van Tol, R.J.P., et al. Structure-based design of novel boronic acid-based inhibitors of autotaxin. Journal of Medicinal Chemistry 54(13), 4619-4626 (2011).
[2] Fulkerson, Z. ,Wu, T.,Sunkara, M., et al. Binding of autotaxin to integrins localizes lysophosphatidic acid production to platelets and mammalian cells. The Journal of Biological Chemisty 286(40), 34654-34663 (2011).
Average Rating: 5 (Based on Reviews and 12 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















