Aldoxorubicin (Synonyms: INNO-206;Doxorubicin-EMCH;INNO 206) |
| رقم الكتالوجGC14858 |
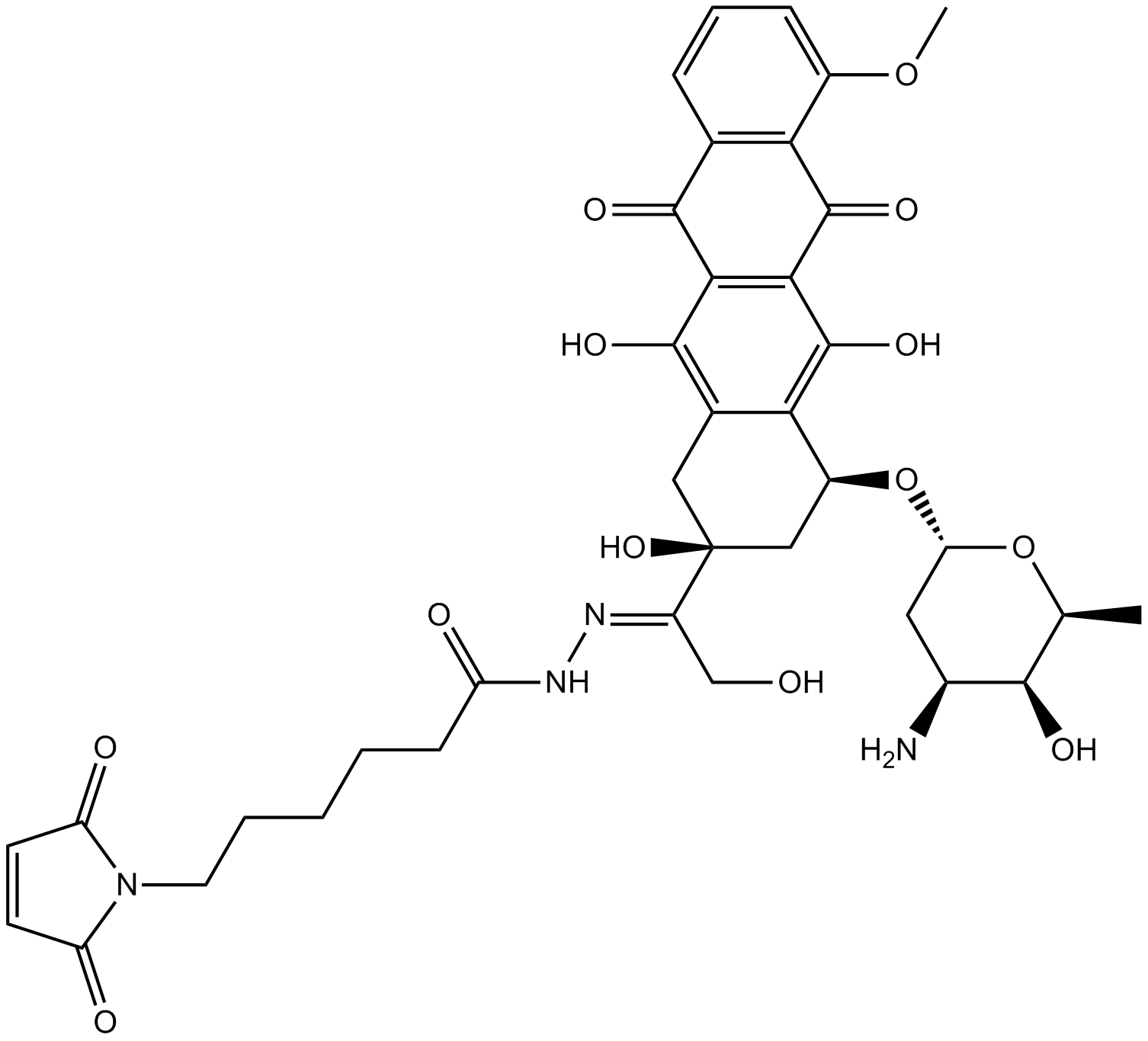
Aldoxorubicin (INNO-206) هو دواء أولي ملزم للألبومين من Doxorubicin (مثبط DNA topoisomerase II) ، والذي يتم إطلاقه من الألبومين تحت الظروف الحمضيةيحتوي Aldoxorubicin (INNO-206) على أنشطة قوية مضادة للأورام في خطوط الخلايا السرطانية المختلفة وفي نماذج أورام الفئران
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 1361644-26-9
Sample solution is provided at 25 µL, 10mM.
Aldoxorubicin (INNO-206) is an albumin-binding prodrug of doxorubicin, which is released from albumin under acidic conditions. Aldoxorubicin (INNO-206) has potent antitumor activities in various cancer cell lines and in murine tumor models.
Aldoxorubicin (INNO-206) (0.27 to 2.16 μM) inhibits blood vessel formation and reduces multiple myeloma cell growth in a pH-dependent fashion[1].
Aldoxorubicin (INNO-206) (10.8 mg/kg, i.v.) shows significantly smaller tumor volumes and IgG levels on days 28, and is well tolerated with 90% of mice surviving until the termination of the study in the mice bearing the LAGκ-1A tumor[1]. Aldoxorubicin (INNO-206) shows a good safety profile at doses up to 260 mg/mL doxorubicin equivalents, and is able to induce tumor regressions in breast cancer, small cell lung cancer and sarcoma in phase I study[2]. Aldoxorubicin (INNO-206) shows superior activity over doxorubicin in a murine renal cell carcinoma model and in breast carcinoma xenograft models[3].
Reference:
[1]. Eric Sanchez, et al. Anti-Myeloma Effects of the Novel Anthracycline Derivative INNO-206. Clin Cancer Res.2012 18; 3856.
[2]. Kratz, F. INNO-206 (DOXO-EMCH), an Albumin-Binding Prodrug of Doxorubicin Under Development for Phase II Studies. Current Bioactive Compounds, 2011, 7(1): 33-38(6)
[3]. Graeser R, et al. INNO-206, the (6-maleimidocaproyl hydrazone derivative of doxorubicin), shows superior antitumor efficacy compared to doxorubicin in different tumor xenograft models and in an orthotopic pancreas carcinoma model. Invest New Drugs. 2010 F
[4]. Walker L, et al. Cell penetrating peptides fused to a thermally targeted biopolymer drug carrier improve the delivery and antitumor efficacy of an acid-sensitive doxorubicin derivative. Int J Pharm. 2012 Oct 15;436(1-2):825-32.
Average Rating: 5 (Based on Reviews and 3 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















