ARP 100 (Synonyms: CAY10609, Matrix Metalloproteinase-2 Inhibitor III, MMP2 Inhibitor III) |
| Catalog No.GC13487 |
A selective inhibitor of MMP-2
Products are for research use only. Not for human use. We do not sell to patients.
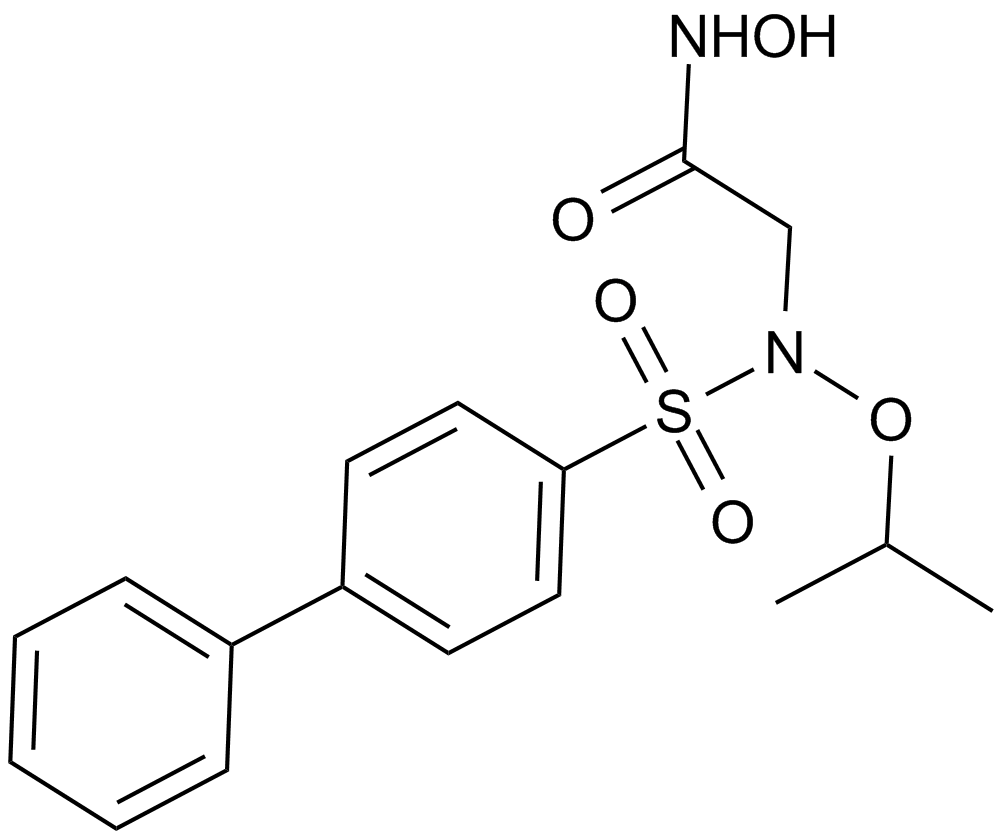
Cas No.: 704888-90-4
Sample solution is provided at 25 µL, 10mM.
ARP 100 is a selective inhibitor of matrix metalloproteinases 2 (MMP-2) with IC50 value of 12 nM [1].
Proteins of the matrix metalloproteinase (MMP) family are involved in the breakdown of extracellular matrix in normal physiological processes, such as embryonic development, reproduction, and tissue remodeling, as well as in disease processes, such as arthritis and metastasis (http://en.wikipedia.org/wiki/MMP2). ARP 100 is a selective inhibitor of matrix metalloproteinases 2 (MMP-2).
In vitro: As a potent derivative of N-arylsulfonyl-N-alkoxyaminoacetohydroxamic acids, ARP 100, shows similar anti-invasive properties to the analogue reference drug CGS27023A, in an in vitro model of invasion on matrigel, carried out on cellular lines of fibrosarcoma HT1080 (tumoural cells over-expressing MMP-2 and MMP-9) [1].
In silico: ARP 100 proved to be practically inactive in the MMP-1; this was probably due to the presence of the biphenyl group, since this substituent was not able to interact in the S10 pocket, and this fact determined the loss of the interaction of the oxygen atoms of the sulfonamido group with Ala182 and Leu181. ARP 100 was also found to be the most selective ligand of the four analyzed, and this fact could be due to the presence of a large P10 group, rigid and unable to form H bonds, or to interact with the MMP-1-S10 pocket. This analysis suggested that in order to maintain the MMP-2/MMP-1 selectivity, the presence of the biphenyl group as a P10 substituent could be very useful [2].
Clinical trial: ARP 100 is currently in the preclinical development and no clinical trial is ongoing.
References:
[1] Rossello A, Nuti E, Orlandini E, Carelli P, Rapposelli S, Macchia M, Minutolo F, Carbonaro L, Albini A, Benelli R, Cercignani G, Murphy G, Balsamo A. New N-arylsulfonyl-N- alkoxyaminoacetohydroxamic acids as selective inhibitors of gelatinase A (MMP-2). Bioorg Med Chem. 2004;12(9):2441-50.
[2] Tuccinardi T, Martinelli A, Nuti E, Carelli P, Balzano F, Uccello-Barretta G, Murphy G, Rossello A.Amber force field implementation, molecular modelling study, synthesis and MMP-1/MMP-2 inhibition profile of (R)- and (S)-N-hydroxy-2-(N-isopropoxybiphenyl-4-ylsulfonamido)- 3-methylbutanamides. Bioorg Med Chem. 2006;14(12):4260-76.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















