Defactinib (Synonyms: PF-04554878, VS-6063) |
| Catalog No.GC16813 |
Le defactinib (VS-6063; PF-04554878) est un nouvel inhibiteur de FAK doté d'activités antiangiogéniques et antinéoplasiques potentielles.
Products are for research use only. Not for human use. We do not sell to patients.
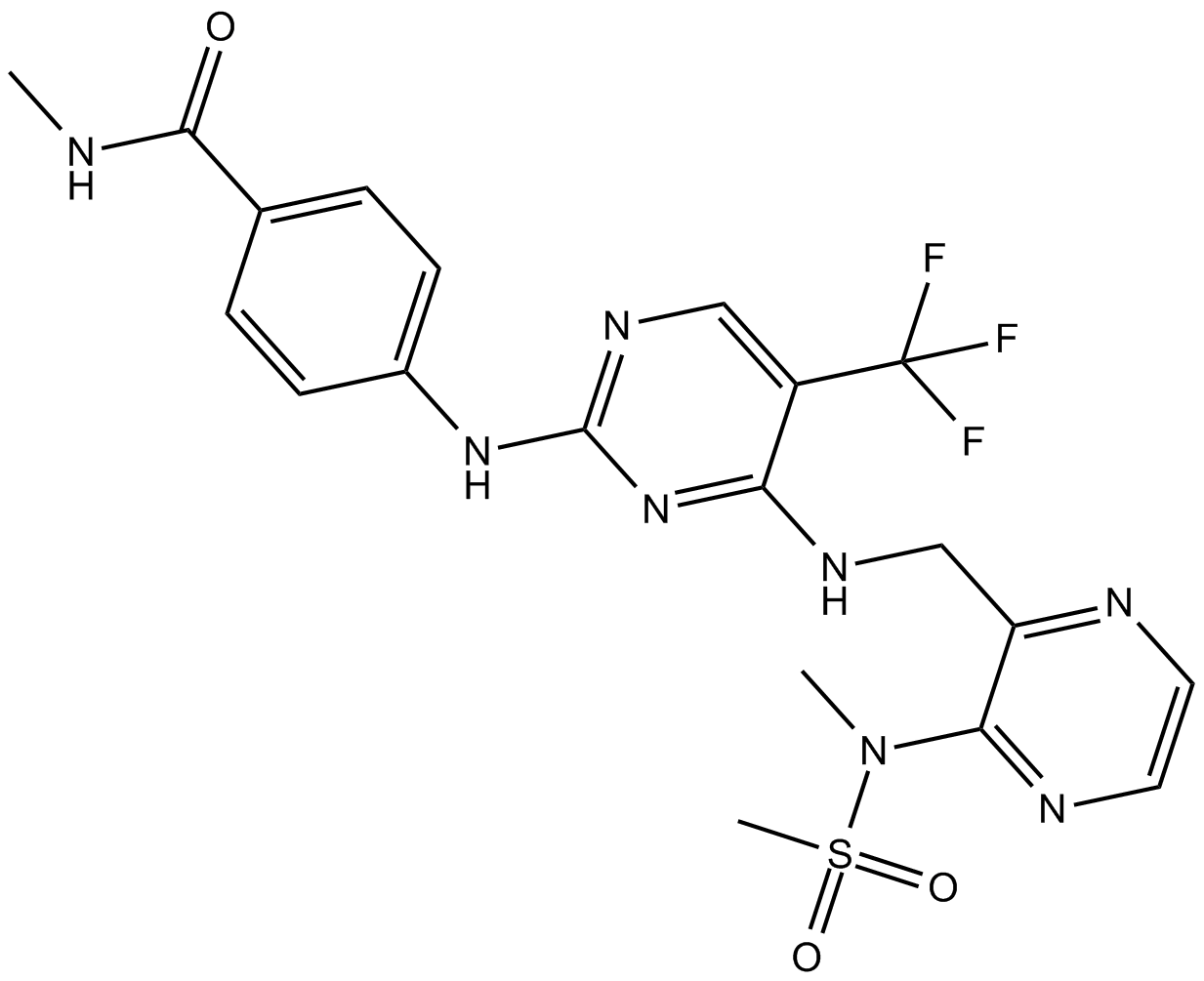
Cas No.: 1073154-85-4
Sample solution is provided at 25 µL, 10mM.
Defactinib (VS-6063; PF-04554878) is a novel FAK inhibitor with potential antiangiogenic and antineoplastic activities.
Defactinib (VS-6063) inhibits FAK phosphorylation at the Tyr397 site in a time- and dose-dependent manner. RPPA data shows that Defactinib reduces levels of AKT and YB-1 in taxane-resistant cell lines. The expression of pFAK (Tyr397) is statistically significantly inhibited by Defactinib in a dose-dependent manner in all cell lines. Defactinib inhibits pFAK (Tyr397) expression within 3 hours, with a gradual return of expression by 48 hours[1].
Defactinib (VS-6063) doses of 25 mg/kg twice a day or greater statistically significantly inhibits pFAK (Tyr397) at 3 hours, with return of expression noted by 24 hours. Therefore, administration of Defactinib at 25 mg/kg twice a day is selected as the dosing schedule for subsequent therapy experiments. For therapy experiments, female nude mice bearing HeyA8 tumors in the peritoneal cavity are randomly divided into 4 groups (n=10 per group): 1) vehicle orally twice daily and phosphate-buffered saline intraperitoneally weekly (control); 2) Defactinib 25 mg/kg orally twice daily; 3) PTX intraperitoneally weekly; and 4) both VDefactinib 25 mg/kg orally twice daily and PTX intraperitoneally weekly. There is an 87.4% reduction in tumor weight by PTX monotherapy in the HeyA8 model, and combination therapy resulted in the greatest tumor weight reduction, with a 97.9% reduction (P=0.05 compared with PTX). In the SKOV3ip1 model, a 92.7% tumor weight reduction is observed in the combination group compared with PTX (P<0.001)[1].
References:
[1]. Kang Y, et al. Role of focal adhesion kinase in regulating YB-1-mediated resistance in ovarian cancer. J Natl Cancer Inst. 2013 Oct 2;105(19):1485-95.
Average Rating: 5 (Based on Reviews and 20 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















