Ladarixin sodium (Synonyms: DF 2156A) |
| Catalog No.GC62296 |
La ladarixine sodique (DF 2156A) est un antagoniste actif par voie orale, allostérique non compétitif et double CXCR1 et CXCR2.
Products are for research use only. Not for human use. We do not sell to patients.
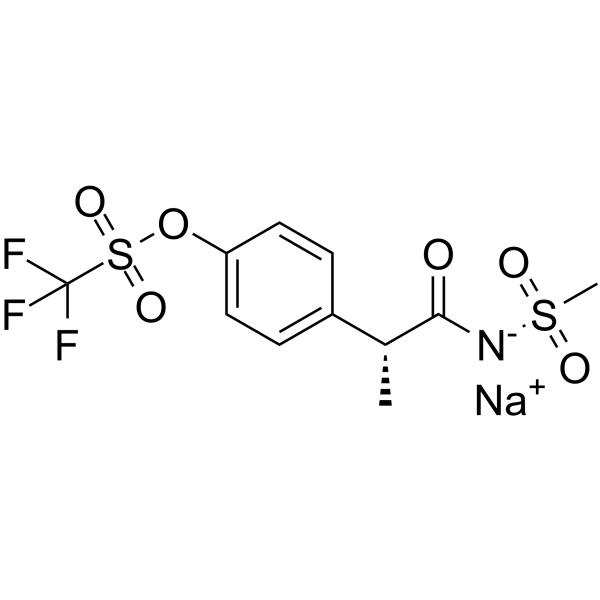
Cas No.: 865625-56-5
Sample solution is provided at 25 µL, 10mM.
Ladarixin sodium (DF 2156A) is an orally active, allosteric non-competitive and dual CXCR1 and CXCR2 antagonist. Ladarixin sodium can be used for the research of COPD and asthma[1].
Ladarixin inhibits human polymorphonuclear leukocyte (PMN) migration to CXCL8 (IC50 at 0.7 nM)[2].
Ladarixin (10 mg/kg; p.o. once a day) reduces allergic airway inflammation in a model of single OVA exposure. Ladarixin reduces allergic airway inflammation, remodeling, and bronchial hyperreactivity in a model of chronic OVA exposure[1].Ladarixin (10 mg/kg; p.o. once a day for 8 days) reduces pulmonary inflammation and fibrosis induced by bleomycin in mice[1].Ladarixin (10 mg/kg; p.o. once a day for 3 days) protects mice from cigarette smoke-induced exacerbation of influenza-A infection[1].Ladarixin is also effective in decreasing CXCL8-induced polymorphonuclear leukocyte infiltration in several animal models without a significant dose-related reduction in systemic neutrophil counts[2].
[1]. Matheus Silverio Mattos, et al. CXCR1 and CXCR2 Inhibition by Ladarixin Improves Neutrophil-Dependent Airway Inflammation in Mice. Front Immunol. 2020 Oct 2;11:566953.
[2]. Daria Marley Kemp, et al. Ladarixin, a dual CXCR1/2 inhibitor, attenuates experimental melanomas harboring different molecular defects by affecting malignant cells and tumor microenvironment. Oncotarget. 2017 Feb 28;8(9):14428-14442.
Average Rating: 5 (Based on Reviews and 11 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















