Oleoyl 3-carbacyclic Phosphatidic Acid (Synonyms: 3ccPA 18:1) |
| Catalog No.GC44500 |
Cyclic phosphatidic acids (cPAs) are naturally occurring analogs of lysophosphatidic acid (LPA) in which the sn-2 hydroxy group forms a 5-membered ring with the sn-3 phosphate.
Products are for research use only. Not for human use. We do not sell to patients.
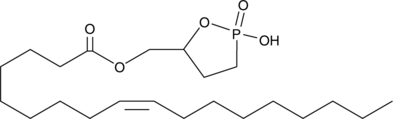
Cas No.: 779333-58-3
Sample solution is provided at 25 µL, 10mM.
Cyclic phosphatidic acids (cPAs) are naturally occurring analogs of lysophosphatidic acid (LPA) in which the sn-2 hydroxy group forms a 5-membered ring with the sn-3 phosphate. Carba-derivatives of cPA (ccPA) are modified at the sn-2 (2-ccPA) or sn-3 (3-ccPA) linkage, preventing the opening of cPA to produce lysophosphatidic acid (LPA). Oleoyl 3-Carbacyclic Phosphatidic Acid (3-ccPA 18:1) is a cyclic LPA analog that contains the 18:1 fatty acid, oleate, at the sn-1 position of the glycerol backbone. At 25 μM, it inhibits the transcellular migration of MM1 cells across mesothelial cell monolayers in response to fetal bovine serum (90.1%) or LPA (99.9%) without affecting proliferation. 3-ccPA 18:1, at 0.1-1.0 μM, significantly inhibits autotaxin, an enzyme that is important in cancer cell survival, growth, migration, invasion, and metastasis.
Average Rating: 5 (Based on Reviews and 13 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















