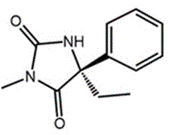
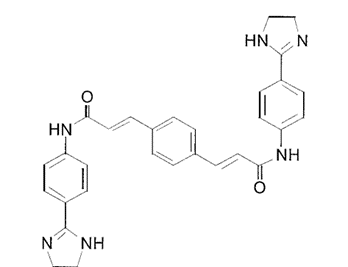
S-Mephenytoin
Chemically, Mephenytoin is 3-methyl-5, 5-phenyl-ethyl-hydantoin that is also known as Mesantoin. Mephenytoin is a molecule, that can be clinically used as a pharmaceutically active drug therapeutic drug. For any pharmaceutically active drug, its structural features are the single primary factor that not only determines the site of biochemical transformation during metabolism but also the rate of that biochemical transformation. The molecular structure of Mephenytoin is shown in Figure 1, while the existence of a chiral carbon at carbon number 5 and its chemical implications in terms of existence of Mephenytoin as a racemic mixture, are diagrammatically demonstrated in Figure 2.
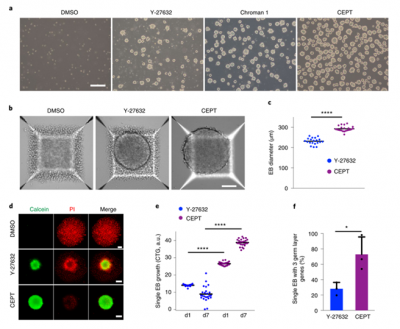
CEPT
Human pluripotent stem cells (HPSCs) have the ability to self-renew, but are highly sensitive to environmental disturbances in vitro, posing a challenge for their therapeutic applications. Further breakthroughs are urgently needed to ensure the safe and stable long-term growth and functional differentiation of these cells. Here, a high-throughput screening strategy is used to identify a small molecule mixture that can enhance the survival ability of hPSCs and their differentiated offspring. The combination of Chroman 1, Emricasan, Polyamines, and trans-ISRIB (CEPT) improves the cell survival rate of genetically stable hPSCs by simultaneously blocking several stress mechanisms that originally damage cell structure and function. CEPT provides powerful improvements for several key applications in stem cell research, including conventional cell passage, cryopreservation of pluripotent stem cells and differentiated cells, embryoid body (EB) and organoid formation, single cell cloning, and genome editing. Therefore, CEPT represents a unique multi pharmacological strategy for comprehensive cell protection, providing a theoretical basis for the efficient and safe utilization of hPSCs.
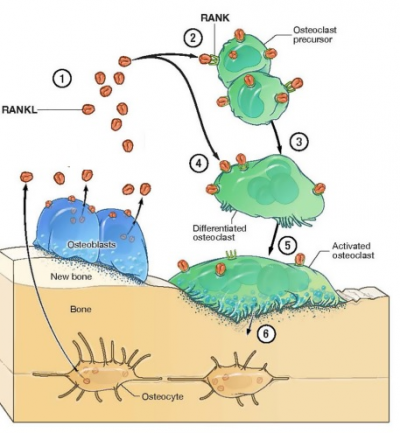
Denosumab
Denosumab is novel pharmaceutical drug. Chemically, it is a monoclonal antibody (mAb), that has the full-length sequence same as of the human antibodies; thus, it is sometimes regarded as fully human. It was initially developed using XenoMouse transgenic mouse technology, which is a powerful tool used for the synthesis of human antibodies in bulk quantity . It selectively and actively binds to the Receptor activator of nuclear factor kappa-Β ligand (RANKL) with a strong interaction. It has a good safety profile as a therapeutical drug against osteoporosis. Other reason that makes Denosumab not only the suitable but also the best treatment option is that it has a longer half live. That essentially means that less frequency of the Denosumab dosage would be required. Less dosage frequency is an important factor when it comes to use any pharmaceutical drug for the therapeutic purposes in the clinical setting.
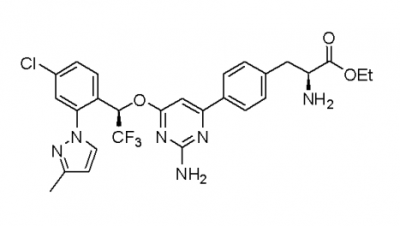
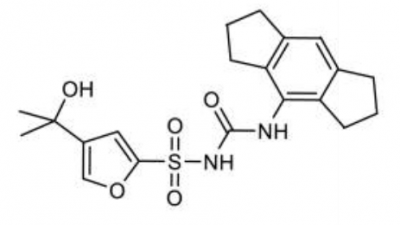
MCC950 Sodium
CP-456773, cytokine release inhibitory drug-3 CRID3, MCC950 sodium and MCC950, all are the short forms for N-(1,2,3,5,6,7-hexahydro-S-indacen-4-ylcarbamoyl)-4-(2-hydroxy-2-propanyl)-2-furansulfonamide. Chemically, it is a derivative of diaryl sulfonylurea derivative. It is a small molecule. Its molecular structure is diagrammatically depicted in Figure 1. The roadmap for its synthesis is documented in the scientific literature in detail.
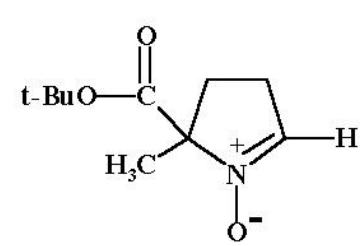
BMPO
BMPO, BocMPO or 5-BMPO cpd is the short form available for 5-tert-butoxycarbonyl 5-methyl-1-pyrroline N-oxide. It is novel spin trap, which is butoxylated. It molecular structure is diagrammatically depicted in Figure 1. The detailed protocol for its synthesis has already been documented in the scientific literature
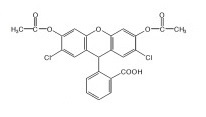
H2DCFDA (DCFH-DA)
H2DCFDA or DCFH-DA is the short form, which is used for 2',7'-dichlorodihydrofluorescein diacetate. Its molecular structure is diagrammatically depicted in Figure 1. It is neutral as it does not carry a net electric charge, thus it is non polar also.
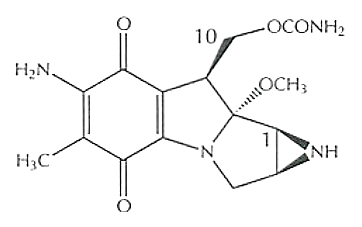
Mitomycin C
Mitomycin C is also known as 7-amino-9α-methoxymitosane (Carlos de Oliveira and Wilson, 2020). Chemically, Mitomycin C is a small molecule. Its molecular structure is diagrammatically shown in Figure 1.
GW 4869
Chemically, GW 4869 is a small and novel molecule highly specific in its inhibitory action. Its molecular structure is diagrammatically depicted in Figure 1. It is cationic and also hydrophobic in its behavior. GW 4869 inhibits sphingomyelinases (SMase), more specifically neutral sphingomyelinase 2 (N-SMase 2), also termed sphingomyelin phosphodiesterase 3 (SMPD3) (Vuckovic et al., 2017) in a non-competitive and Magnesium ion dependent manner under both conditions, i.e., in vitro and in vivo, with the half-maximal inhibitory concentration (IC 50) of 1 micro-Molar (μM). This inhibition is tissue dependent as GW 4869 does not inhibit neutral sphingomyelinase 2 (N-SMase 2) in the Multiple myeloma (MM) cells (Vuckovic et al., 2017). SMase is a hydrolase thus, when active it catalyzes the hydrolysis of Sphingomyelin that results in the production of phosphorylcholine and lipid ceramide, that is bioactive in nature. As this hydrolysis occurs optimally at the pH value 7, thus these SMases are termed as neutral sphingomyelinase (N-SMase) (Canals et al., 2011).
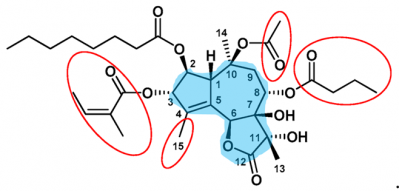
Thapsigargin
Thapsigargin
Thapsigargin (shortened as Tg) is a small yet structurally complex molecule, that inhibits the sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) at the concentration of nano Molar. Tg at this concentration is particularly safe for the other transmembrane pumps It is the extensively studied SERCA inhibitor. The structure of carbon backbone in the Tg is very complex as there lies a fusion of three rings in the heart of its molecular structure.