CP21 |
| Catalog No.GC48954 |
An iron chelator
Products are for research use only. Not for human use. We do not sell to patients.
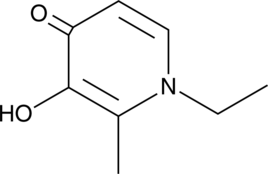
Cas No.: 30652-12-1
Sample solution is provided at 25 µL, 10mM.
CP21 is an iron chelator that binds to iron in a 3:1 (ligand:iron) ratio.1 It is active against P. falciparum when used at concentrations of 10 and 100 µM.2 CP21 inhibits production of prostaglandin I2 induced by epinephrine, arachidonic acid , or A23187 in isolated rat aortic rings with IC50 values of 1.3, 1.3, and 1.4 mM, respectively.3 It inhibits glutamate-induced oxytosis, as well as decreases iodoacetic acid-induced cytotoxicity in an in vitro model of ischemia, in HT22 mouse hippocampal cells (EC50s = 13 and 9.5 µM, respectively).4 CP21 (200 mg/kg) increases the excretion of iron, but not copper, zinc, calcium, or magnesium, in rabbits.5
1.Dobbin, P.S., Hider, R.C., Hall, A.D., et al.Synthesis, physicochemical properties, and biological evaluation of N-substituted 2-alkyl-3-hydroxy-4(1H)-pyridinones: Orally active iron chelators with clinical potentialJ. Med. Chem.36(17)2448-2458(1993) 2.Heppner, D.G., Hallaway, P.E., Kontoghiorghes, G.J., et al.Antimalarial properties of orally active iron chelatorsBlood72(1)358-361(1988) 3.Jeremy, J.Y., Kontoghiorghes, G.J., Hoffbrand, A.V., et al.The iron chelators desferrioxamine and 1-alkyl-2-methyl-3-hydroxypyrid-4-ones inhibit vascular prostacyclin synthesis in vitroBiochem. J.254(1)239-244(1988) 4.Maher, P., and Kontoghiorghes, G.J.Characterization of the neuroprotective potential of derivatives of the iron chelating drug deferiproneNeurochem. Res.40(3)609-620(2015) 5.Kontoghiorghes, G.J., and Hoffbrand, A.V.Orally active α-ketohydroxy pyridine iron chelators intended for clinical use: In vivo studies in rabbitsBr. J. Haematol.62(4)607-613(1986)
Average Rating: 5 (Based on Reviews and 37 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















