L-3-n-Butylphthalide
Drugs including calcium channel blockers, excitatory amino acid receptor antagonists and free radical scavengers are used for the prevention and treatment of ischemic CVDs such as strokes. Such drugs have side effects as well.
2,3,5,4′- tetrahydroxystilbene-2-O-β-D- glucopyranoside (TSG)
2,3,5,4′- tetrahydroxystilbene-2-O-β-D- glucopyranoside (TSG)(GC26316), identified as the most prevalent bioactive constituent derived from Polygonum multiflorum (Heshouwu), a traditional Chinese herb. TSG is a polyphenol compound that implies numerous antioxidative, anti-aging, anti-inflammatory, anti-hypercholesterolemic, hepatoprotective, and anti-tumor aspects of pharmacological actions. TSG was primarily found to be distributed in the liver after oral administration by investigating the distribution of Polygonum multiflorum characteristic elements in animal models. Moreover, comparing treated PM with raw PM indicates that raw PM exhibits much more severe hepatotoxicity than treated one, assuming that a higher concentration of TSG in raw PM probably be a toxic constituent of PM.
1-Deoxynojirimycin (DNJ)
1-Deoxynojirimycin (DNJ) chemical formula C6H13NO4 and molecular weight of 163.17 g/mol, is a type of indigenous alkaloid examined in mulberry leaves, insects, and the culture broth of some microbes as a product of secondary metabolites such as Commelina communis, and bacterial strains including Bacillus and Streptomyces species. As a representative iminopyranose, 1-Deoxynojirimycin exhibits α-glucosidase inhibitor activity along with anti-viral, anti-diabetic, anti-inflammatory, anti-obesity, and antioxidant properties employed for several applications.
Pembrolizumab
Pembrolizumab, marketed under the brand name Keytruda, is a monoclonal antibody used in cancer immunotherapy. It targets the programmed cell death protein 1 (PD-1) receptor on T cells, a critical checkpoint in the immune system.
β-Nicotinamide mononucleotide
With respect to longevity and enhanced health, scientists have turned their attention towards a remarkable molecule: Beta Nicotinamide Mononucleotide (NMN). It’s a derivative of Vitamin B3 (niacin), has gained attention for its potential to modulate cellular function, combat the aging, and promote overall well-being. β-Nicotinamide mononucleotide (NMN) is a natural product which exists in small quantity in most plants, such as edamame, broccoli, and cucumber.
Atezolizumab (MPDL3280A)
Atezolizumab is an Fc-engineered , humanized, monoclonal antibody that targets programmed death ligand 1 (PD-L1). Atezolizumab prevents the binding of PD-L1 to receptors programmed death 1 (PD-1) and B7.
3-Methyladenine
By searching deep in the field of cellular biology by using the microscopic aspects, we revealed the surprising molecule under the name of 3-Methyladenine (3-MA), a compound that does not have its recognization under the heading of DNA or proteins, but play a vital role in the front line of cellular processes.
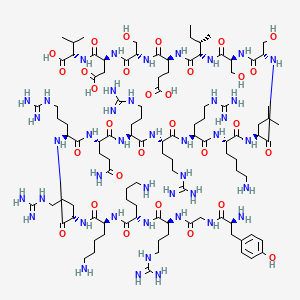
Tat-NR2B9c
In the ever-evolving landscape of biomedical research, a new star is rising, capturing the attention of scientists and researchers worldwide. This star is none other than Tat-NR2B9c, a peptide whose unique properties and potential applications are setting the stage for groundbreaking advancements in medicine and therapeutic treatment.
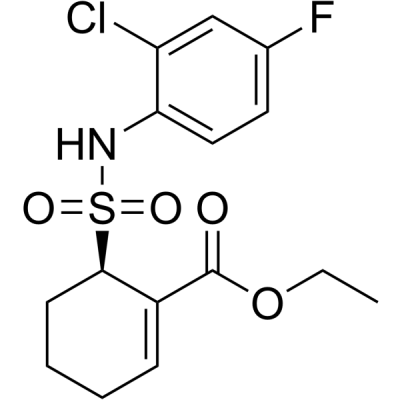
TAK-242
TAK-242 ethyl (6R)-6-[N-(2-chloro-4-fluorophenyl)sulfamoyl]cyclohex-1-ene-1-carboxylate, also known as resatorvid, is a cyclohexene derivative and a small inhibitor molecule of TLR4 signaling, which was first represented as a novel anti-sepsis treatment that may inhibit the production of inflammatory mediators. Research on the mechanisms of action has revealed that TAK-242 specifically binds to Cys747 in the TIR domain of TLR4, which ensuingly prevents TLR4 from interacting with TIRAP.