Fosaprepitant |
| カタログ番号GC16246 |
Fosaprepitant (L-785298) は、Aprepitant のプロドラッグです。
Products are for research use only. Not for human use. We do not sell to patients.
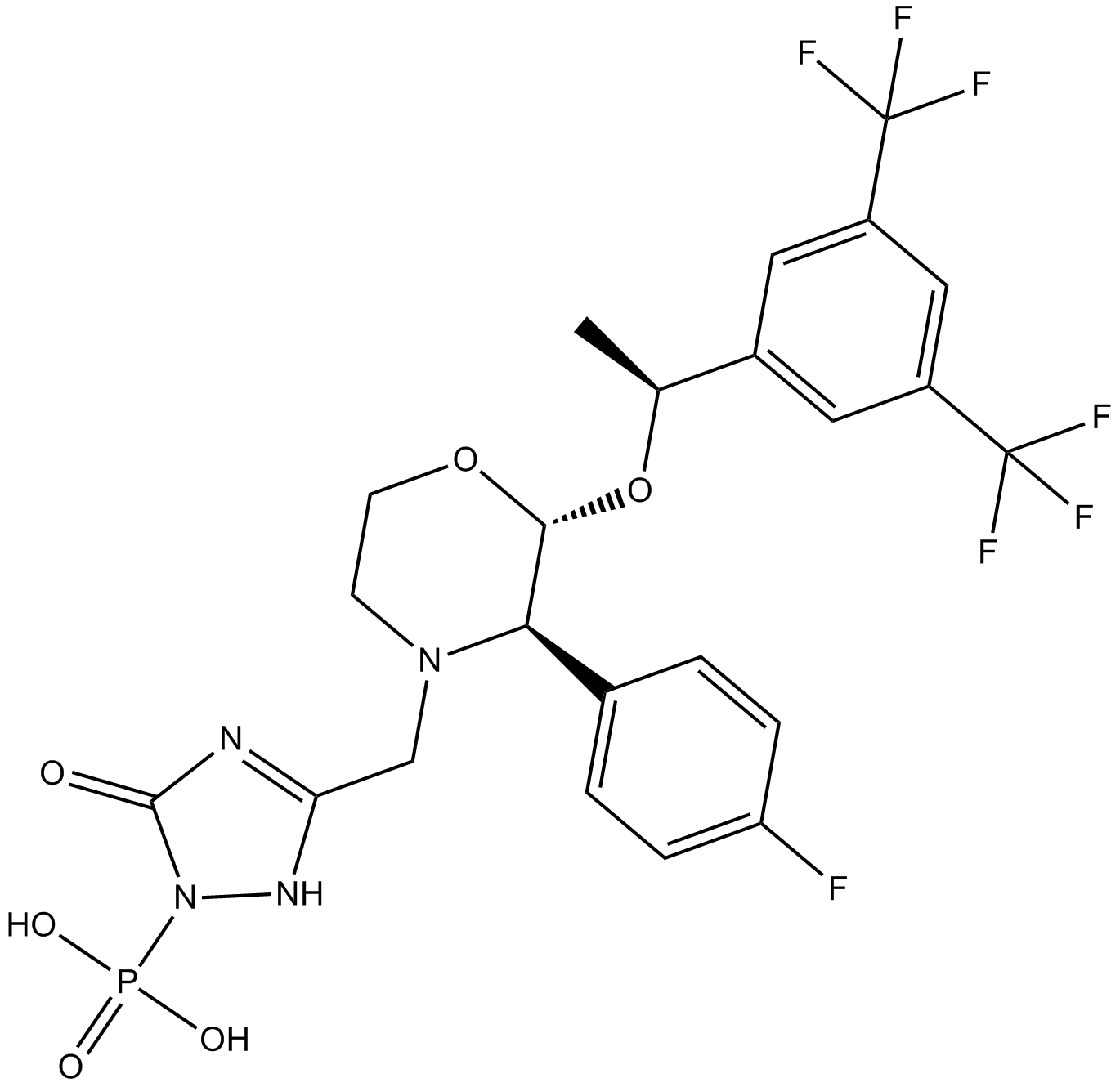
Cas No.: 172673-20-0
Sample solution is provided at 25 µL, 10mM.
Fosaprepitant (L-758,298 MK-0517) is an antagonist of neurokinin-1[1].
Fosaprepitant has shown the pharmacodynamic function by its active metabolite aprepitant. Fosaprepitanot has been found to be a highly selective antagonist of the NK-1 receptor and inhibit the cation of substance P. In addition, Fosaprepitant has been exhibited to have the effect on cisplatin induced emesis in the classical ferret model. Besides, because of the brain penetrating of aprepitant, Fosaprepitant has been revealed to have a very high affinity for the NK-1 receptor and increase the efficacy by dexamethasone, granisetron and so on. Fosaprepitant has been evaluated to use in the prevention of chemotherapy-induced nausea and vomiting (CINV) by combination with a 5-HT3 antagonist and a steroid [1].
References:
[1] Van Belle SJ1, Cocquyt V. Fosaprepitant dimeglumine (MK-0517 or L-785,298), an intravenous neurokinin-1 antagonist for the prevention of chemotherapy induced nausea and vomiting.Expert Opin Pharmacother. 2008 Dec; 9(18):3261-70.
Average Rating: 5 (Based on Reviews and 36 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















