JNJ4796 |
| カタログ番号GC65968 |
JNJ4796 は、ヘマグルチニン (HA) を介した融合を阻害することにより、インフルエンザ A グループ 1 ウイルスを中和する、インフルエンザ ウイルスの経口活性融合阻害剤です。 JNJ4796 は、広域中和抗体 (bnAbs) の機能を模倣しています。
Products are for research use only. Not for human use. We do not sell to patients.
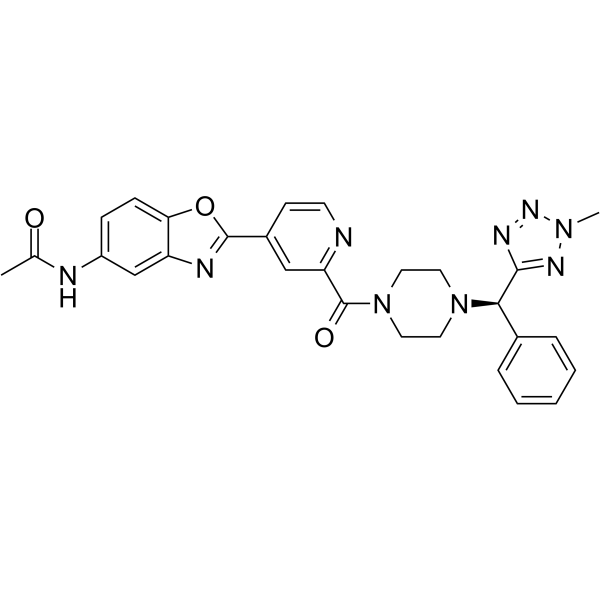
Cas No.: 2241664-16-2
Sample solution is provided at 25 µL, 10mM.
JNJ4796 is an oral active fusion inhibitor of influenza virus, neutralizing influenza A group 1 viruses by inhibiting hemagglutinin (HA)-mediated fusion. JNJ4796 mimics the functionality of the broadly neutralizing antibodies (bnAbs)[1].
Like bnAb CR6261, the mechanism of action of JNJ4796 is demonstrated to be based on inhibition of the pH-sensitive conformational change of HA that triggers fusion of the viral and endosomal membranes and release of the viral genome into the host cell[1].
Oral administration of JNJ4796 protects mice from lethal challenge of 25 times the median lethal dose (LD50) of H1N1 A/Puerto Rico/8/1934 virus. Doses of 50 and 10 mg/kg of JNJ4796 twice daily, initiated one day before challenge and continuing for 7 days, results in 100% survival at day 21 in comparison to the less potent compound JNJ8897 for which less than 50% survival is achieved[1].
Oral doses of JNJ4796 results in dose-dependent efficacy after a sublethal viral challenge (LD90), with twice daily administration of 15 and 5 mg/kg of JNJ4796 giving rise to 100% survival[1].
| Animal Model: | Female BALB/cAnNCrl mice intranasally infected with 2 × 25 μL of 25 × LD50 or 1 × LD90 of H1N1 A/Puerto Rico/8/34 dissolved in sterile phosphate buffered saline (D-PBS)[1] |
| Dosage: | 50 and 10 mg/kg. |
| Administration: | Oral twice daily for 7 days. |
| Result: | Resulted in 100% survival at day 21 in comparison to the less potent compound JNJ8897. |
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















