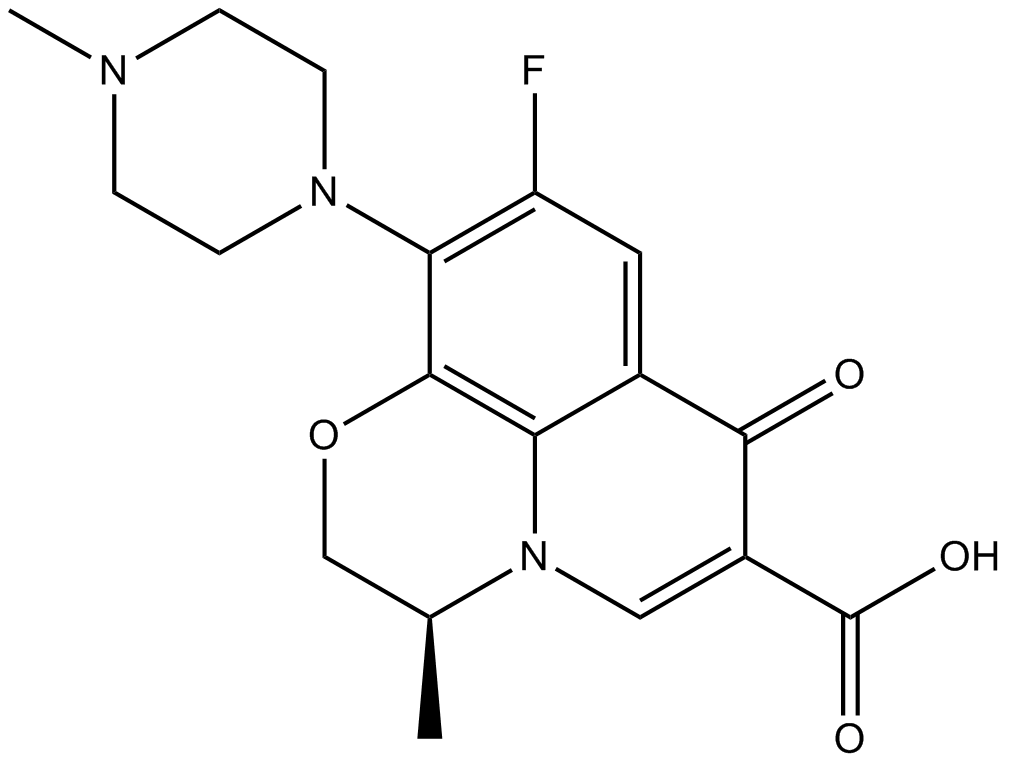
Levofloxacin (Synonyms: MP-376) |
| カタログ番号GC13861 |
レボフロキサシン ((-)-オフロキサシン) は、経口で活性な抗生物質であり、グラム陽性菌とグラム陰性菌の両方に対して活性があります。
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 100986-85-4
Sample solution is provided at 25 µL, 10mM.
Levofloxacin, a synthetic fluoroquinolone, is an antibacterial agent that inhibits the supercoiling activity of bacterial DNA gyrase, halting DNA replication.Target: AntibacterialLevofloxacin reduced bacterial load compared with placebo by 4.9-fold (95% confidence interval, 1.4-25.7; P=0.02) at day 7 but had no effect at any point on any marker of neutrophilic airway inflammation. In patients with a baseline bacterial load of more than 10(6) cfu/mL, levofloxacin treatment was associated with a 26.5% (95% confidence interval, 1.8%-51.3%; P=0.04) greater reduction in the percentage neutrophil count compared with placebo at day 7 [1]. Levofloxacin was found to significantly improve the clinical and microbiological parameters in CP individuals [2]. A 30-day course of levofloxacin does not significantly improve BK viral load reduction or allograft function when used in addition to overall reduction of immunosuppression [3].
References:
[1]. Siva, R., et al., Effect of levofloxacin on neutrophilic airway inflammation in stable COPD: a randomized, double-blind, placebo-controlled trial. Int J Chron Obstruct Pulmon Dis, 2014. 9: p. 179-86.
[2]. Pradeep, A.R., et al., Clinical and microbiological effects of levofloxacin in the treatment of chronic periodontitis: a randomized, placebo-controlled clinical trial. J Investig Clin Dent, 2014.
[3]. Lee, B.T., et al., Efficacy of Levofloxacin in the Treatment of BK Viremia: A Multicenter, Double-Blinded, Randomized, Placebo-Controlled Trial. Clin J Am Soc Nephrol, 2014.
Average Rating: 5 (Based on Reviews and 10 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




