TD52 dihydrochloride |
| カタログ番号GC64936 |
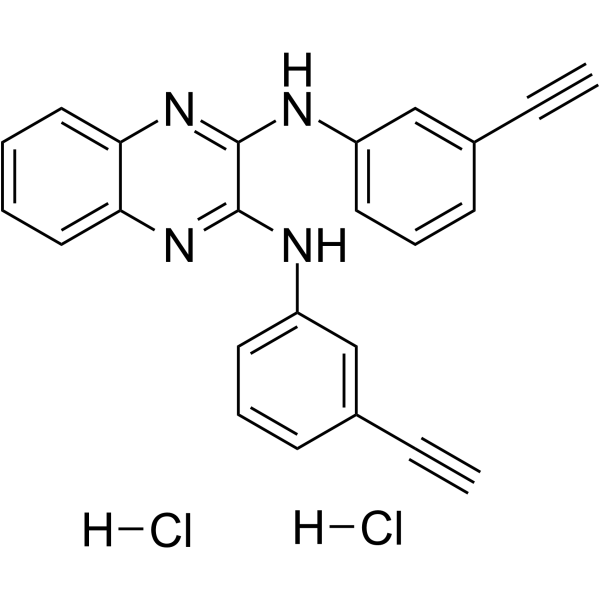
エルロチニブ誘導体である TD52 二塩酸塩は、タンパク質ホスファターゼ 2A (CIP2A) 阻害剤の経口活性で強力な癌性阻害剤です。 TD52 二塩酸塩は、CIP2A/PP2A/p-Akt シグナル伝達経路の調節を介して、トリプルネガティブ乳がん (TNBC) 細胞のアポトーシス効果を媒介します。 TD52 二塩酸塩は、Elk1 の CIP2A プロモーターへの結合を妨害することにより、CIP2A を間接的に減少させました。 TD52 二塩酸塩は p-EGFR 阻害が少なく、強力な抗がん作用があります。
Products are for research use only. Not for human use. We do not sell to patients.
Sample solution is provided at 25 µL, 10mM.
TD52 dihydrochloride, an Erlotinib derivative, is an orally active, potent cancerous inhibitor of protein phosphatase 2A (CIP2A) inhibitor. TD52 dihydrochloride mediates the apoptotic effect in triple-negative breast cancer (TNBC) cells via regulating the CIP2A/PP2A/p-Akt signalling pathway. TD52 dihydrochloride indirectly reduced CIP2A by disturbing Elk1 binding to the CIP2A promoter. TD52 dihydrochloride has less p-EGFR inhibition and has potent anti-cancer activity[1].
TD52 dihydrochloride (2-10 μM; 48 hours) shows anti-proliferative ability and induces differential apoptotic effects in these cell lines[1]. TD52 dihydrochloride (5 μM; 48 hours) has minimal effects on p-EGFR or EGFR expression but downregulated CIP2A expression[1]. TD52 dihydrochloride (2.5, 5, 7.5 μM; 48 hours) time-dependently induces apoptosis accompanied with downregulating CIP2A and p-Akt[1]. TD52 dihydrochloride (5 μM; 24 hours) significantly increases the phosphatase activity of PP2A in TNBC cells[1]. TD52 dihydrochloride (5 μM; 48 hours) has no obvious effects on other common RTKs, such as IGFR, PDGFR and VEGFR2[1].
TD52 dihydrochloride (10 mg/kg/day; oral gavage; for 52 days) significantly inhibits MDA-MB-468 xenograft tumour size and tumour weight[1].
[1]. Chun-Yu Liu, et al. EGFR-independent Elk1/CIP2A signalling mediates apoptotic effect of an erlotinib derivative TD52 in triple-negative breast cancer cells. Eur J Cancer. 2017 Feb;72:112-123.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















