Roblitinib (Synonyms: Roblitinib) |
| Catalog No.GC19154 |
Roblitinib(FGF-401)은 1.9nM의 IC50을 갖는 경구 활성 및 고도로 선택적인 FGFR4 억제제입니다. 로블리티닙은 항종양 활성이 있습니다.
Products are for research use only. Not for human use. We do not sell to patients.
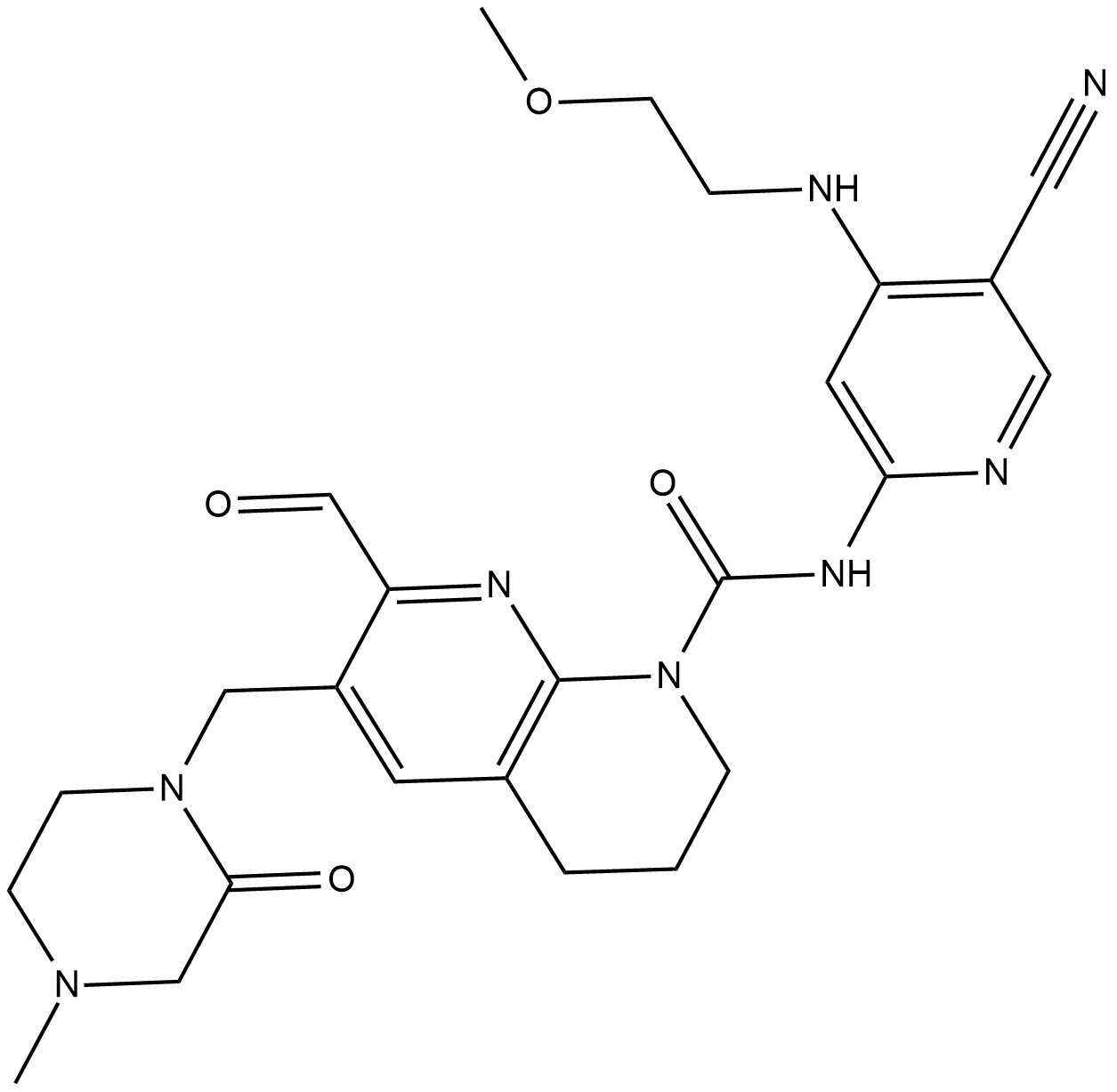
Cas No.: 1708971-55-4
Sample solution is provided at 25 µL, 10mM.
Roblitinib (FGF-401) is a 1,8-naphthyridine pyridine derivative[5].Roblitinib binds to an inactive (autoinhibited brake, closed activation segment) DFG-Din enzyme form; the ligand binds covalently to FGFR4 and is classified as a Type VI inhibitor[7].
Roblitinib as an orally active and highly selective FGFR4 inhibitor with an IC50 of 1.9 nM without off-target effects[3]. Roblitinib has antitumor activity[4]
In mice, PKM2-IN-1 treatment markedly decreased the tumor volume and tumor weight, compared with the control group. Meanwhile, no significant weight reduction was detected in the mouse treated with PKM2-IN-1, suggesting that PKM2-IN-1 did not cause any major organ toxicity. Thus, use of specific PKM2 inhibitors to block the glycolytic pathway and target cancer cell metabolism represents a promising therapeutic approach for treating PKM2-overexpressing ovarian cancer[6].In hearts of 7-day-old mice, PKM2-specific inhibitor PKM2-IN-1 significantly blocked the proliferation of cardiomyocytes in HRR groups, indicating HRR-induced proliferation of cardiomyocytes was fully abolished by PKM2-IN-1[2]
References:
[1]: Zou Y, Zheng S, et,al. N6-methyladenosine regulated FGFR4 attenuates ferroptotic cell death in recalcitrant HER2-positive breast cancer. Nat Commun. 2022 May 13;13(1):2672. doi: 10.1038/s41467-022-30217-7. PMID: 35562334; PMCID: PMC9106694.
[2]: Chan SL, Schuler M, et,al. A first-in-human phase 1/2 study of FGF401 and combination of FGF401 with spartalizumab in patients with hepatocellular carcinoma or biomarker-selected solid tumors. J Exp Clin Cancer Res. 2022 Jun 2;41(1):189. doi: 10.1186/s13046-022-02383-5. PMID: 35655320; PMCID: PMC9161616.
[3]: National Center for Biotechnology Information (2022). PubChem Patent Summary for US-9266883-B2. Retrieved September 6, 2022 from https://pubchem.ncbi.nlm.nih.gov/patent/US-9266883-B2.
[4]: Fairhurst RA, Knoepfel T, et,al. Discovery of Roblitinib (FGF401) as a Reversible-Covalent Inhibitor of the Kinase Activity of Fibroblast Growth Factor Receptor 4. J Med Chem. 2020 Nov 12;63(21):12542-12573. doi: 10.1021/acs.jmedchem.0c01019. Epub 2020 Oct 1. PMID: 32930584.
[5]: Roskoski R Jr. The role of fibroblast growth factor receptor (FGFR) protein-tyrosine kinase inhibitors in the treatment of cancers including those of the urinary bladder. Pharmacol Res. 2020 Jan;151:104567. doi: 10.1016/j.phrs.2019.104567. Epub 2019 Nov 23. PMID: 31770593.
[6]: Zhou Z , Chen X , et,al. Characterization of FGF401 as a reversible covalent inhibitor of fibroblast growth factor receptor 4. Chem Commun (Camb). 2019 May 21;55(42):5890-5893. doi: 10.1039/c9cc02052g. PMID: 31041948.
[7]: Roskoski R Jr. Classification of small molecule protein kinase inhibitors based upon the structures of their drug-enzyme complexes. Pharmacol Res. 2016 Jan;103:26-48. doi: 10.1016/j.phrs.2015.10.021. Epub 2015 Oct 31. PMID: 26529477.
[8]: Weiss A, Adler F, et,al. FGF401, A First-In-Class Highly Selective and Potent FGFR4 Inhibitor for the Treatment of FGF19-Driven Hepatocellular Cancer. Mol Cancer Ther. 2019 Dec;18(12):2194-2206. doi: 10.1158/1535-7163.MCT-18-1291. Epub 2019 Aug 13. PMID: 31409633.
Average Rating: 5 (Based on Reviews and 9 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















