PF-06380101 (Synonyms: Aur0101; Auristatin-0101) |
| Katalog-Nr.GC33136 |
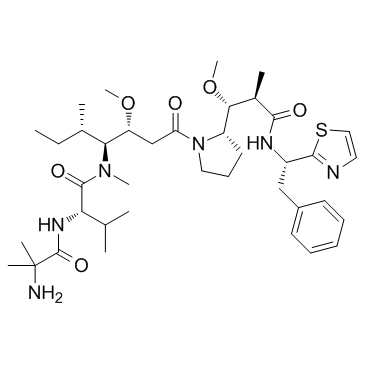
PF-06380101 (Aur0101), ein Auristatin-Mikrotubuli-Inhibitor, ist ein zytotoxisches Dolastatin-10-Analogon. PF-06380101 (Aur0101) zeigt im Vergleich zu anderen synthetischen Auristatin-Analoga, die bei der Herstellung von ADCs verwendet werden, hervorragende Potenzen in Tumorzellproliferationsassays und unterschiedliche ADME-Eigenschaften.
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 1436391-86-4
Sample solution is provided at 25 µL, 10mM.
PF-06380101 is a novel cytotoxic Dolastatin 10 analogue; with excellent potencies in tumor cell proliferation assays and differential ADME properties when compared to other synthetic auristatin analogues that are used in the preparation of ADCs.IC50 value: ~0.2 nM(GI50 in BT474, MDA-MB-361-DYT2 and N87 cell line) [1]Target: ADCs cytotoxin; tubulin inhibitorAfter an IV dose of 20a at 20 μg/kg to Wistar Han rats, PF-06380101 exhibited a mean systemic clearance (Cl) of 70 mL/min/kg and a volume of distribution (Vss) of 14.70 L/kg, resulting in a terminal elimination half-life (t1/2) of approximately 6 h. PF-06380101 preferentially distributes into human plasma relative to whole blood and that PF-06380101 is a P-glycoprotein (P-gp) substrate. PF-06380101 is anticipated to be of low risk to perpetrate pharmacokinetic drug interactions with compounds for which CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and/or CYP3A4/5-mediated metabolism constitutes the primary mechanism of clearance. The utility of the new auristatin analogues as ADC payloads including the development of the lead analogue 20a (PF-06380101) will be reported in due course.
[1]. Maderna A, et al. Discovery of cytotoxic dolastatin 10 analogues with N-terminal modifications. J Med Chem. 2014 Dec 26;57(24):10527-43.
Average Rating: 5 (Based on Reviews and 19 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















