U 46619 (Synonyms: 9,11-dideoxy-9α,11α-methanoepoxy PGF2α, 9,11-dideoxy-9α,11α-methanoepoxy Prostaglandin F2α) |
| Katalog-Nr.GC13205 |
Selektiver Agonist des PGH2/TxA2 (TP) Rezeptors
Products are for research use only. Not for human use. We do not sell to patients.
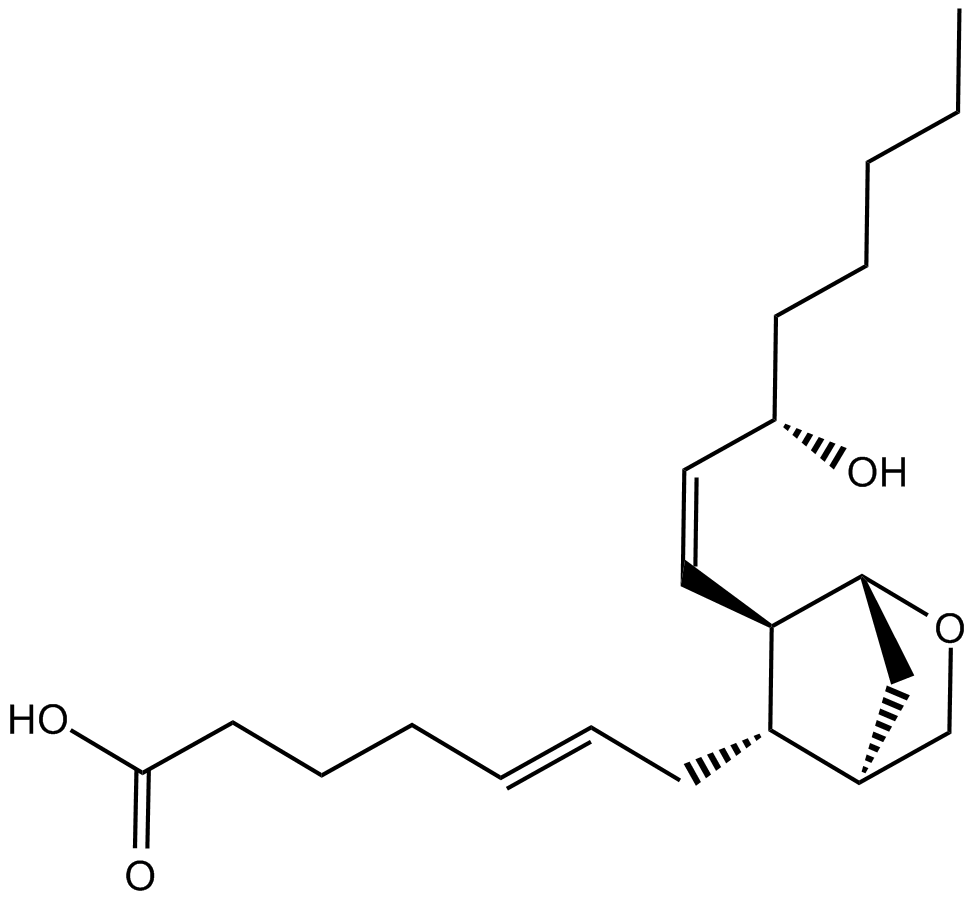
Cas No.: 56985-40-1
Sample solution is provided at 25 µL, 10mM.
EC50: 0.035 μM for shape change, 0.057 μM for MLCP, 0.536 μM for serotonin release, 1.31 μM for aggregation and 0.53 μM for fibrinogen receptor binding [3].
Prostaglandin endoperoxide analogue U46619 (11,9 epoxymethano-prostaglandin H2) is a selective agonist of prostaglandin H2 (PGH2)/thromboxane A2 (TxA2) (TP) receptor. Both PGH2 and TxA2 receptors are G-protein coupled receptors.
In vitro: U46619 showed a biphasic effect on human platelets. Shape change and MLCP occurred at low concentrations of this compound (EC50 = 0.035 uM and 0.057 uM), whereas serotonin release, platelet at higher concentrations (EC50 = 0.536 uM and 1.31 uM). The effect on platelet shape change and MLCP is receptor mediated [3].
In vivo:. U46619, through activation of ETA and ETB receptors, elicits renal cortical vasoconstriction and medullary vasodilation in the rat [2]. In conscious SHR (spontaneously hypertensive rats), 1-100 nmol/kg U-46619 (i.c.v.) induced a dose-related increase in blood pressure but had no significant effect on heart rate [1].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Sirén AL, Svartstrm-Fraser M, Paakkari I. Central cardiovascular effects of the endoperoxide analogue U-46619 in rats. Prostaglandins Leukot Med. 1985 Mar;17(3):381-6.
[2] Hantz H, Adesuyi A, Adebayo O. Differential effects of U46619 on renal regional hemodynamics in the rat: involvement of endothelin. J Pharmacol Exp Ther. 2001 Oct;299(1):372-6.
[3] Morinelli TA1, Niewiarowski S, Daniel JL, Smith JB. Receptor-mediated effects of a PGH2 analogue (U 46619) on human platelets. Am J Physiol. 1987 Nov;253(5 Pt 2):H1035-43.
Average Rating: 5 (Based on Reviews and 9 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *





