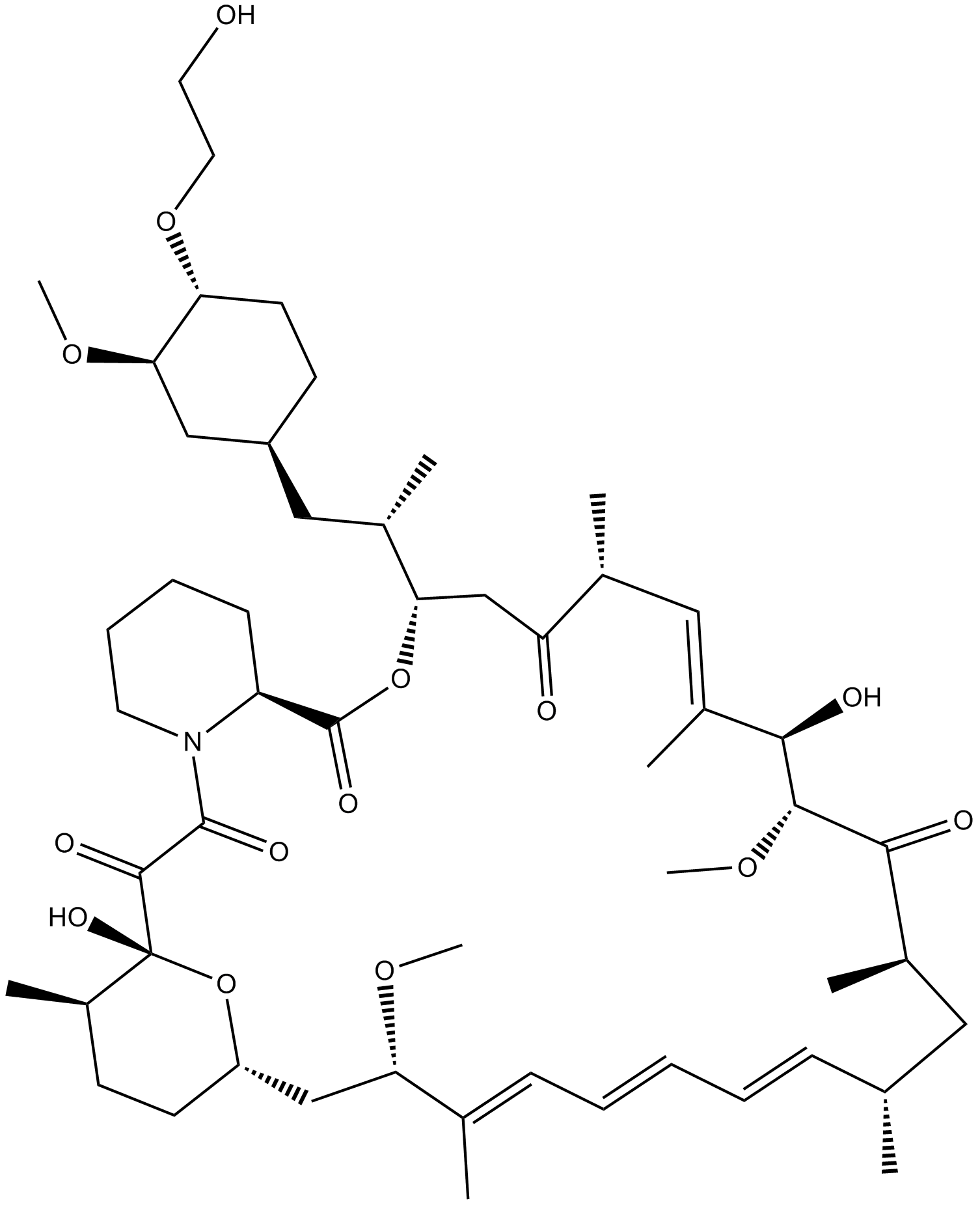
Everolimus (RAD001) (Synonyms: RAD001) |
| カタログ番号GC13601 |
ラパマイシン誘導体
Products are for research use only. Not for human use. We do not sell to patients.
Cas No.: 159351-69-6
Sample solution is provided at 25 µL, 10mM.
Everolimus (RAD001) is an orally active derivative of rapamycin that inhibits the Ser/Thr kinase, mTOR (mammalian target of rapamycin).[1]
In vitro activity of everolimus it displayed that the dose-dependent inhibition of cell growth by everolimus using methylene blue staining after 96 hours of incubation in four different human tumor cell lines, which can be regarded as sensitive (HCT-15, A549) and insensitive (KB-31 and HCT-116).[1] In vitro efficacy test, antiproliferative concentrations of RAD001 resulted in total dephosphorylation of S6K1 and the substrate S6 and a shift in the mobility of 4E-BP1, with IC50 of 0.7 nmol/L and 1,778 nmol/L in both the sensitive murine B16/BL6 melanoma and the insensitive human cervical KB-31,respectively.[2] In vitro study, combination gemcitabine (100 nM) with everolimus (0.05-2 μM) had significantly antiproliferative effect with an arrest of cell cycle at S phase.[3]
In vivo experimental it shown that everolimus is very well tolerated with no obvious clinical signs of toxicity; even when treating for up to 60 mg/kg per day by oral gavage the maximum tolerated dosage was not reached. In vivo efficacy study, daily orally treatment with everolimus (0.5 or 2.5 mg/kg) dose-dependently inhibited growth, and using a higher dose of 5 mg/kg once or twice per week also showed similar antitumor efficacy in the rat CA20498 model.[1] In vivo, treatment with 0.1-10 mg/kg/d RAD001 dose-dependently increased the hemoglobin content but reduced the Tie-2 content and this was significant for VEGF stimulation but not bFGF stimulation.[2]
References:
[1].O'Reilly T, McSheehy PM. Biomarker Development for the Clinical Activity of the mTOR Inhibitor Everolimus (RAD001): Processes, Limitations, and Further Proposals. Transl Oncol. 2010 Apr;3(2):65-79.
[2].Lane HA, et al. mTOR inhibitor RAD001 (everolimus) has antiangiogenic/vascular properties distinct from a VEGFR tyrosine kinase inhibitor. Clin Cancer Res. 2009 Mar 1;15(5):1612-22.
[3].Pinto-Leite R, et al. Everolimus enhances gemcitabine-induced cytotoxicity in bladder-cancer cell lines. J Toxicol Environ Health A. 2012;75(13-15):788-99.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




