BHBM |
| Catalog No.GC46100 |
An acylhydrazone with antifungal activity
Products are for research use only. Not for human use. We do not sell to patients.
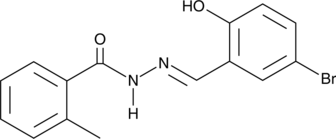
Cas No.: 302807-84-7
Sample solution is provided at 25 µL, 10mM.
BHBM is an acylhydrazone with antifungal activity.1,2 It is active against C. neoformans in vitro (MIC80 = 1 μg/ml).2 BHBM (0.25, 1, and 4 μg/ml) inhibits the synthesis of glucosylceramide, which is essential to fungal cell division, in C. neoformans but not J774 murine macrophages.1 In vivo, BHBM (1.2 mg/kg per day) increases survival in a mouse model of C. neoformans infection.
|1. Haranahalli, K., Lazzarini, C., Sun, Y., et al. SAR studies on aromatic acylhydrazone-based inhibitors of fungal sphingolipid synthesis as next-generation antifungal agents. J. Med. Chem. 62(17), 8249-8273 (2019).|2. Lazzarini, C., Haranahalli, K., Rieger, R., et al. Acylhydrazones as antifungal agents targeting the synthesis of fungal sphingolipids. Antimicrob. Agents Chemother. 62(5), e00156-00118 (2018).
Average Rating: 5 (Based on Reviews and 2 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















