Sorafenib
|
| カタログ番号GC17369 |
ソラフェニブは、Raf-1とB-Rafをターゲットとするマルチキナーゼ阻害剤であり、それぞれのIC50値は6 nMおよび22 nMです。
Products are for research use only. Not for human use. We do not sell to patients.
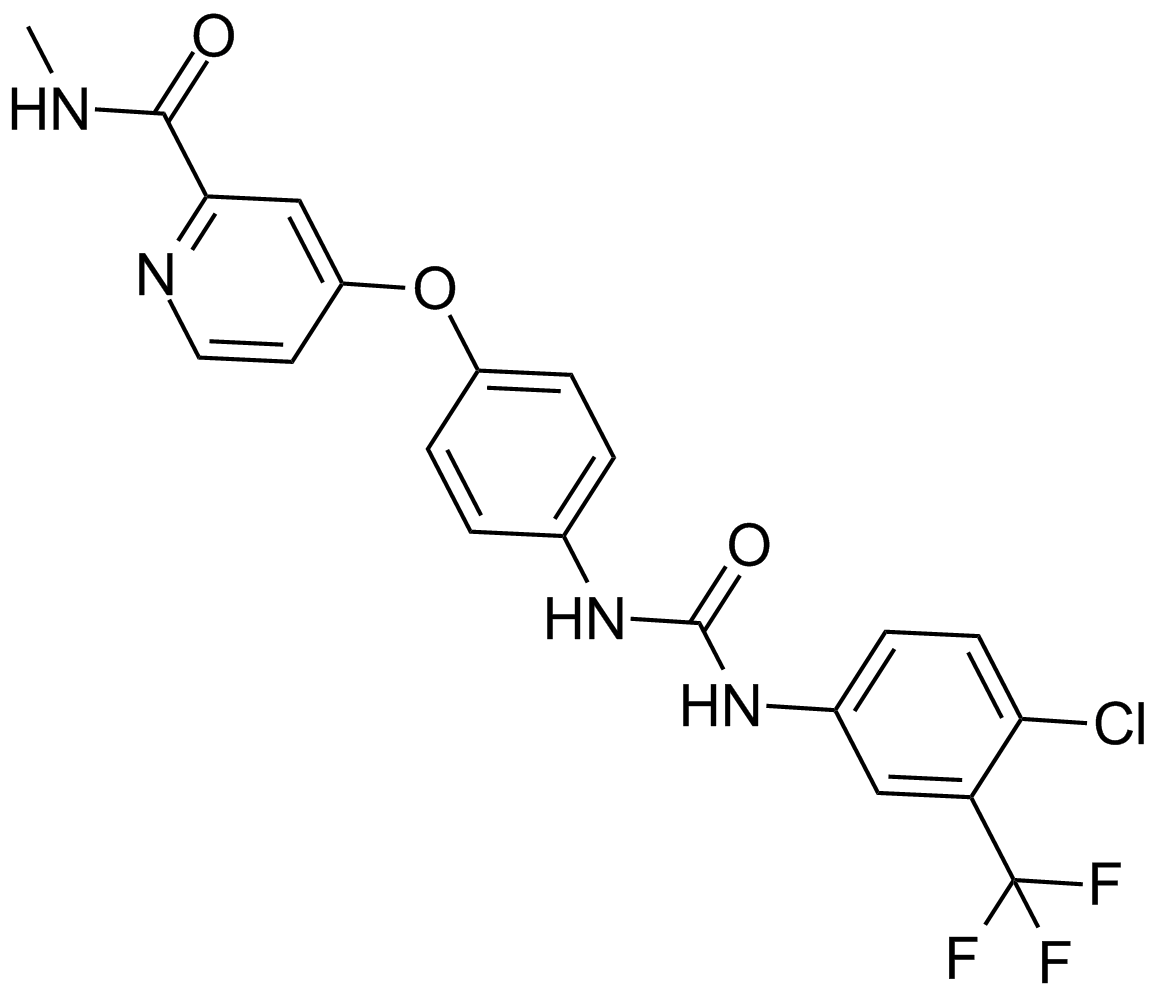
Cas No.: 284461-73-0
Sample solution is provided at 25 µL, 10mM.
Sorafenib acts as a multi-kinase inhibitor, targeting Raf-1 and B-Raf with IC50 values of 6 nM and 22 nM, respectively. Additionally, Sorafenib demonstrates inhibitory effects on VEGFR-2, VEGFR-3, PDGFR-β, Flt-3, and c-KIT, displaying corresponding IC50 values of 90 nM, 20 nM, 57 nM, 59 nM, and 68 nM. Beyond these kinase activities, Sorafenib is capable of inducing autophagy and apoptosis while triggering ferroptosis activation, resulting in its notable antitumor efficacy [1-3].
Sorafenib(5-40μM; 24 h) had a dose-dependent inhibitory effect on HSC-T6 cells viability [4]. Sorafenib(25mM;0-42h) alters the lipid composition in Huh7.5 cells[5].
Sorafenib(2.5, 5, 10 mg/kg; i.p; twice a week for 8 weeks) attenuated liver injury and extracellular matrix (ECM) accumulation in CCl4 -induced fibrotic livers, accompanied by reduction of SLC7A11 and GPX4 proteins[4]. Treatment with erastin and sorafenib(10 mg/kg; i.p; once every other day) alleviated liver fibrosis in mice by inducing hepatic stellate cells(HSCs) ferroptosis in mice[6].The synergism of sorafenib and T cells is mediated via reduced ATF4 expression, causing activation of the IRF7-IL-15 axis in leukemia cells and thereby leading to metabolic reprogramming of leukemia-reactive T cells in humans[7].
References:
[1]. Abdelgalil AA, Alkahtani HM, et,al.Sorafenib. Profiles Drug Subst Excip Relat Methodol. 2019;44:239-266. doi: 10.1016/bs.podrm.2018.11.003. Epub 2019 Jan 18. PMID: 31029219.
[2]. Wilhelm SM, Carter C, et,al.BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004 Oct 1;64(19):7099-109. doi: 10.1158/0008-5472.CAN-04-1443. PMID: 15466206.
[3]. Xia S, Pan Y, et,al.The microenvironmental and metabolic aspects of sorafenib resistance in hepatocellular carcinoma. EBioMedicine. 2020 Jan;51:102610. doi: 10.1016/j.ebiom.2019.102610. Epub 2020 Jan 6. PMID: 31918403; PMCID: PMC7000339.
[4]. Yuan S, Wei C, et,al. Sorafenib attenuates liver fibrosis by triggering hepatic stellate cell ferroptosis via HIF-1α/SLC7A11 pathway. Cell Prolif. 2022 Jan;55(1):e13158. doi: 10.1111/cpr.13158. Epub 2021 Nov 22. PMID: 34811833; PMCID: PMC8780895.
[5]. Liu G, Kuang S, et,al.Sorafenib kills liver cancer cells by disrupting SCD1-mediated synthesis of monounsaturated fatty acids via the ATP-AMPK-mTOR-SREBP1 signaling pathway. FASEB J. 2019 Sep;33(9):10089-10103. doi: 10.1096/fj.201802619RR. Epub 2019 Jun 14. PMID: 31199678.
[6]. Zhang Z, Guo M, et,al. RNA-binding protein ZFP36/TTP protects against ferroptosis by regulating autophagy signaling pathway in hepatic stellate cells. Autophagy. 2020 Aug;16(8):1482-1505. doi: 10.1080/15548627.2019.1687985. Epub 2019 Nov 11. PMID: 31679460; PMCID: PMC7469536.
[7]. Mathew NR, Baumgartner F,et,al. Sorafenib promotes graft-versus-leukemia activity in mice and humans through IL-15 production in FLT3-ITD-mutant leukemia cells. Nat Med. 2018 Mar;24(3):282-291. doi: 10.1038/nm.4484. Epub 2018 Feb 12. Erratum in: Nat Med. 2018 Apr 10;24(4):526. PMID: 29431743; PMCID: PMC6029618.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *





