Definitions of commonly used pharmacological terms
Term |
Description |
| Agonist | A drug that binds to and activates a receptor. Can be full, partial or inverse. A full agonist has high efficacy, producing a full response while occupying a relatively low proportion of receptors. A partial agonist has lower efficacy than a full agonist. It produces sub-maximal activation even when occupying the total receptor population, therefore cannot produce the maximal response, irrespective of the concentration applied. An inverse agonist produces an effect opposite to that of an agonist, yet binds to the same receptor binding-site as an agonist. |
| Allosteric Modulator | A drug that binds to a receptor at a site distinct from the active site. Induces a conformational change in the receptor, which alters the affinity of the receptor for the endogenous ligand. Positive allosteric modulators increase the affinity, whilst negative allosteric modulators decrease the affinity. |
| Antagonist | A drug that attenuates the effect of an agonist. Can be competitive or non-competitive, each of which can be reversible or irreversible. A competitive antagonist binds to the same site as the agonist but does not activate it, thus blocks the agonist's action. A non-competitive antagonist binds to an allosteric (non-agonist) site on the receptor to prevent activation of the receptor. A reversible antagonist binds non-covalently to the receptor, therefore can be "washed out". An irreversible antagonist binds covalently to the receptor and cannot be displaced by either competing ligands or washing. |
| Bmax | The maximum amount of drug or radioligand, usually expressed as picomoles (pM) per mg protein, which can bind specifically to the receptors in a membrane preparation. Can be used to measure the density of the receptor site in a particular preparation. |
| Cheng-Prusoff Equation | Used to determine the Ki value from an IC50 value measured in a competition radioligand binding assay: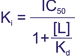 Where [L] is the concentration of free radioligand, and Kd is the dissociation constant of the radioligand for the receptor. |
| Desensitization | A reduction in response to an agonist while it is continuously present at the receptor, or progressive decrease in response upon repeated exposure to an agonist. |
| EC50 | The molar concentration of an agonist that produces 50% of the maximum possible response for that agonist. |
| ED50 | In vitro or in vivo dose of drug that produces 50% of its maximum response or effect. |
| Efficacy | Describes the way that agonists vary in the response they produce when they occupy the same number of receptors. High efficacy agonists produce their maximal response while occupying a relatively low proportion of the total receptor population. Lower efficacy agonists do not activate receptors to the same degree and may not be able to produce the maximal response. |
| Ex vivo | Taking place outside a living organism. |
| Half-life | Half-life (t½) is an important pharmacokinetic measurement. The metabolic half-life of a drug in vivo is the time taken for its concentration in plasma to decline to half its original level. Half-life refers to the duration of action of a drug and depends upon how quickly the drug is eliminated from the plasma. The clearance and distribution of a drug from the plasma are therefore important parameters for the determination of its half-life. |
| i.a. | Intra-arterial route of drug administration. |
| IC50 | In a functional assay, the molar concentration of an agonist or antagonist which produces 50% of its maximum possible inhibition. In a radioligand binding assay, the molar concentration of competing ligand which reduces the specific binding of a radioligand by 50%. |
| i.c. | Intracerebral route of drug administration. |
| i.c.v. | Intracerebroventricular route of drug administration. |
| ID50 | In vitro or in vivo dose of a drug that causes 50% of the maximum possible inhibition for that drug. |
| KB | The equilibrium dissociation constant for a competitive antagonist: the molar concentration that would occupy 50% of the receptors at equilibrium. |
| Kd | The dissociation constant for a radiolabeled drug determined by saturation analysis. It is the molar concentration of radioligand which, at equilibrium, occupies 50% of the receptors. |
| Ki | The inhibition constant for a ligand, which denotes the affinity of the ligand for a receptor. Measured using a radioligand competition binding assay, it is the molar concentration of the competing ligand that would occupy 50% of the receptors if no radioligand was present. It is calculated from the IC50 value using the. |
| Non-Specific Binding | The proportion of radioligand that is not displaced by other competitive ligands specific for the receptor. It can be binding to other receptors or proteins, partitioning into lipids or other things. |
| pA2 | Measure of the potency of an antagonist. It is the negative logarithm of the molar concentration of an antagonist that would produce a 2-fold shift in the concentration response curve for an agonist. |
| pD2 | The negative logarithm of the EC50 or IC50 value. |
| pEC50 | The negative logarithm of the EC50 value. |
| pIC50 | The negative logarithm of the IC50 value. |
| pKB | The negative logarithm of the KB value. |
| pKd | The negative logarithm of the Kd value. |
| pKi | The negative logarithm of the Ki value. |
| Potency | A measure of the concentrations of a drug at which it is effective. |
| Specific Binding | The proportion of radioligand that can be displaced by competitive ligands specific for the receptor. |
| Silent Antagonist | A drug that attenuates the effects of agonists or inverse agonists, producing a functional reduction in signal transduction. Effects only ligand-dependent receptor activation and displays no intrinsic activity itself. Also known as a neutral antagonist. |


