Tenalisib (RP6530) (Synonyms: RP6530) |
| Catalog No.GC32828 |
Tenalisib (RP6530) (RP6530) is a novel, potent, and selective PI3Kδ and PI3Kγ inhibitor with IC50 values of 25 and 33 nM, respectively.
Products are for research use only. Not for human use. We do not sell to patients.
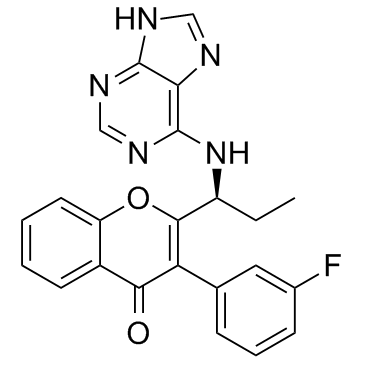
Cas No.: 1639417-53-0
Sample solution is provided at 25 µL, 10mM.
Tenalisib (RP6530) is a novel, potent, and selective PI3Kδ and PI3Kγ inhibitor with IC50 values of 25 and 33 nM, respectively.
Tenalisib shows selectivity over PI3K α (>300-fold) and β (>100-fold) isoforms. Tenalisib exhibits modest proliferation inhibition (33-46% inhibition @ 10 μM) in both HEL-RS and HEL-RR cells. Addition of 10 μM tenalisib to ruxolitinib is synergistic resulting in a near-complete inhibition of proliferation (>90% for HEL-RS and >70% for HEL-RR). Addition of 5 μM tenalisib, 4 h prior to the addition of ruxolitinib results in a significant reduction in EC50of ruxolitinib (5.8 μM) in HEL-RR cells. Incubation of 10 μM tenalisib with ruxolitinib for 72 h increases the percent of apoptotic cells (55% in HEL-RS and 37% in HEL-RR) compared to either agent alone (16-27% in HEL-RS and 17-21% in HEL-RR)[1].
Tenalisib has been well tolerated in subjects with heavily pre-treated relapsed/refractory hematologic malignancies. Reported toxicities are manageable with no DLTs. Single agent activity is evident in difficult-to-treat subjects at ≥ 200 mg BID[2].
[1]. Vakkalanka S, et al. RP6530, a dual PI3K δ/γ inhibitor, potentiates ruxolitinib activity in the JAK2-V617F mutant erythroleukemia cell lines. [abstract]. In: Proceedings of the 106th Annual Meeting of the American Association for Cancer Research; 2015 Apr 18-22; Philadelphia, PA. Philadelphia (PA): AACR; Cancer Res 2015;75(15 Suppl):Abstract nr 2704. doi:10.1158/1538-7445.AM2015-2704 [2]. Carmelo C, et al. A Dose Escalation Study of RP6530, a Novel Dual PI3K Delta/Gamma Inhibitor, in Patients with Relapsed/Refractory Hematologic Malignancies. Blood 2015 126:1495;
Average Rating: 5 (Based on Reviews and 22 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















