Atrasentan hydrochloride |
| Catalog No.GC11783 |
Products are for research use only. Not for human use. We do not sell to patients.
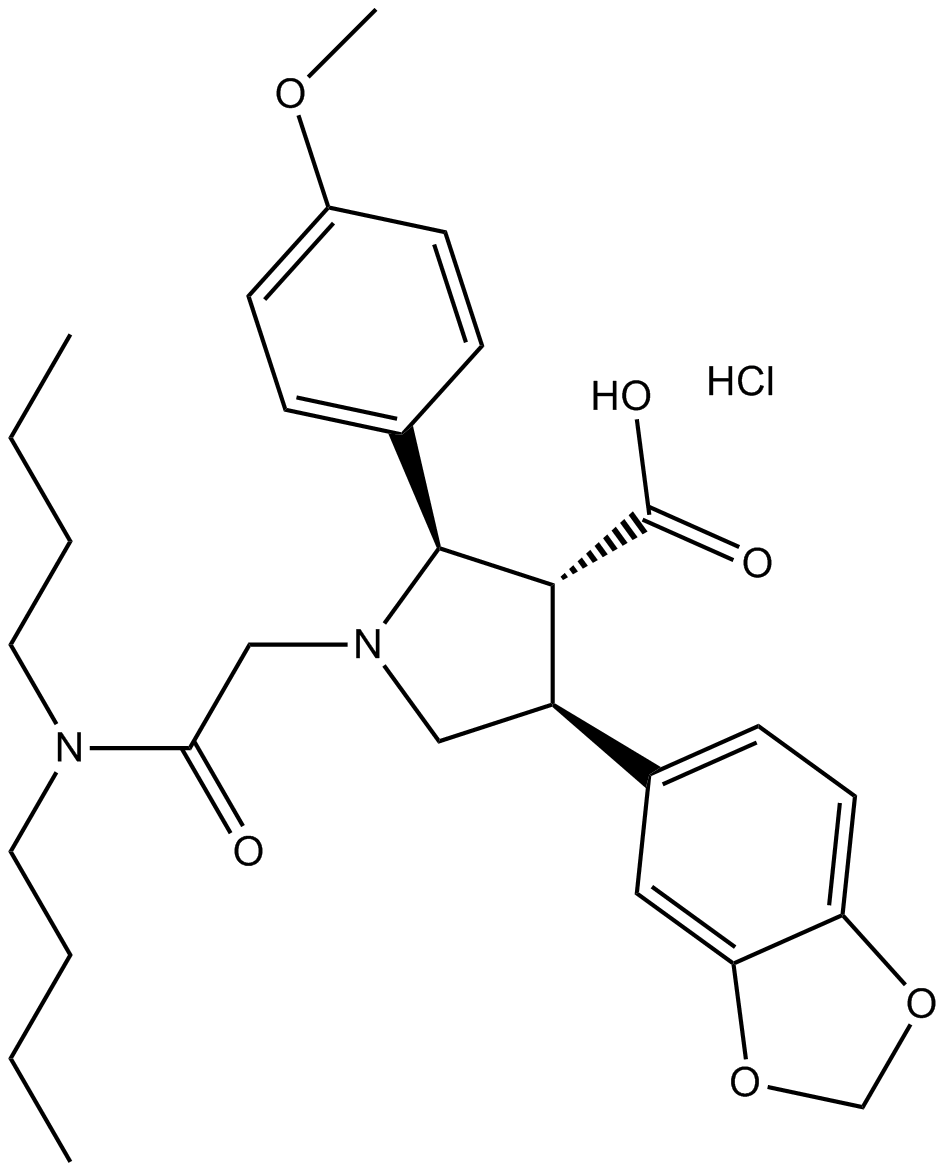
Cas No.: 195733-43-8
Sample solution is provided at 25 µL, 10mM.
Description:
IC50 Value: 0.0551 nM (for ET A receptor) [1]
Atrasentan (A-147627) is an endothelin receptor antagonist being developed at Abbott Laboratories for the treatment of prostate cancer.
in vitro: The combination of Atrasentan with Taxotere was more effective in the inhibition of cell viability and induction of apoptosis in LNCaP and C4-2b cells (androgen receptor positive) but not in PC-3 cells[2]. Atrasentan profoundly induced several CYPs and drug transporters (e.g. 12-fold induction of CYP3A4 at 50 μM). It was a moderate P-gp inhibitor (IC(50) in P388/dx cells = 15.1 ± 1.6 μM) and a weak BCRP inhibitor (IC(50) in MDCKII-BCRP cells = 59.8 ± 11 μM). BCRP or P-gp overexpressing cells were slightly more resistant towards antiproliferative effects of atrasentan [5].
in vivo: ABT-627 did reduce the accumulation of macrophages in both stains (36 to 53%) whereas it blocked by 76% the influx of eosinophils in Balb/c but not in C57Bl/6 mice [3]. Atrasentan was administered orally via drinking water at 3 mg kg-1 per day over 28 days. All diabetic mice developed similar hyperglycaemia (27-30 mmol l-1). Atrasentan treatment significantly improved left ventricular systolic and diastolic function in response to exogenous norepinephrine, but there were no differences between genotypes [4].
Clinical trial: Atrasentan and Zometa for Men With Prostate Cancer Metastatic to Bone . Phase2
Review for Atrasentan hydrochloride
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
Review for Atrasentan hydrochloride
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















