BMS-690514 |
| Catalog No.GC13873 |
Products are for research use only. Not for human use. We do not sell to patients.
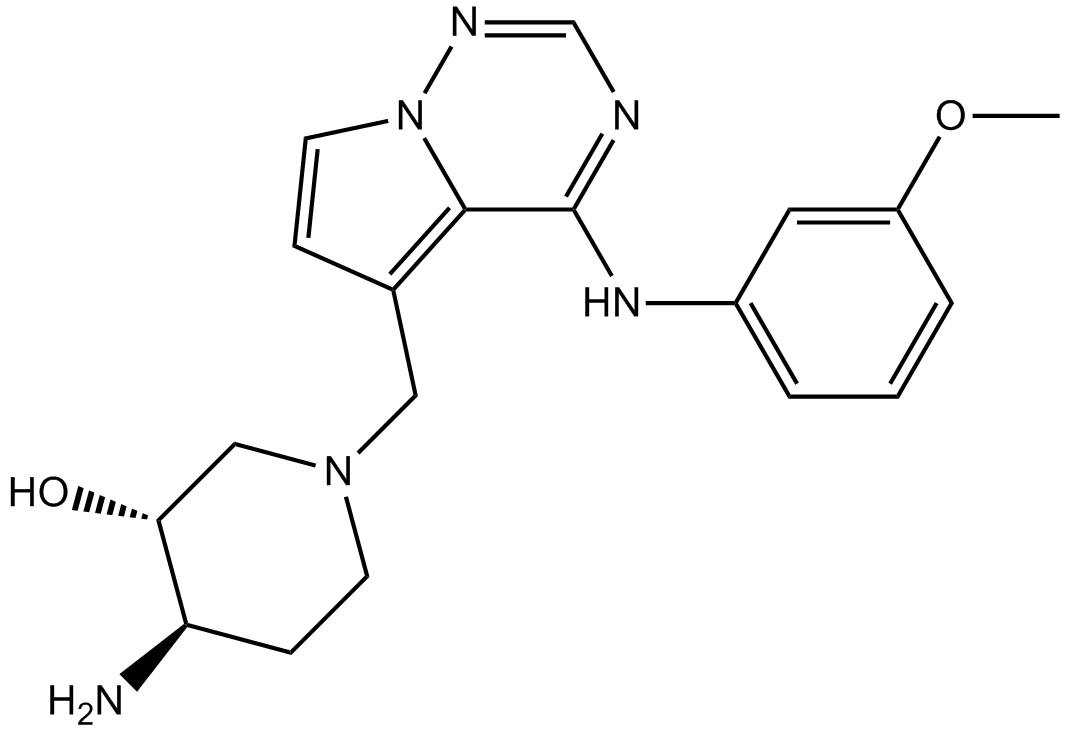
Cas No.: 859853-30-8
Sample solution is provided at 25 µL, 10mM.
BMS-690514, a potent inhibitor of human epidermal growth factor receptor (HER/EGFR). In addition to an improved potency in inhibiting HER1/HER2 with IC50 value of 5 and 19 nM, respectively, BMS-690514 also shows significant potency against other protein kinases, such as VEGFR2, Flt-3, and Lck with IC50 value of 50, 110, and 220 nM, respectively.[1]
The epidermal growth factor receptor is a member of the ErbB family of receptors, EGFR (ErbB-1), HER2/c-neu (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4) [2]. Upon activation by its growth factor ligands, EGFR undergoes a transition from an inactive monomeric form to an active homodimer[3]. In addition to forming homodimers after ligand binding, EGFR may pair with another member of the ErbB receptor family, such as ErbB2/Her2/neu, to create an activated heterodimer. EGFR dimerization stimulates its intrinsic intracellular protein-tyrosine kinase activity, resulting autophosphorylation[4], which elicits downstream activation and signaling by several other proteins with SH2 domains that can initiate several signal transduction cascades, principally the MAPK, Akt and JNK pathways, leading to DNA synthesis and cell proliferation.[5]
Mutations affecting EGFR expression or activity could result in cancer.[6] BMS-690514 is an oral oncologic agent being developed for the treatment of patients with advanced nonsmall cell lung cancer and other solid tumor, for inhibiting the EGFR tyrosine kinase, which is on the cytoplasmic side of the receptor. Without kinase activity, EGFR is unable to activate itself, which is a prerequisite for binding of downstream adaptor proteins. Ostensibly by halting the signaling cascade in cells that rely on this pathway for growth, tumor proliferation and migration is diminished. BMS-690514 is metabolized via multiple metabolic pathways, including P450-mediated oxidation at one of the carbons of its pyrrolotriazine group at this site results in the formation of two metabolites, M1 and M37, through HPLC, NMR, LC/MS/MS and radiochromatographic analysis[7].
References:
1.Punit Marathe. et al. Preclinical Pharmacokinetics and In Vitro Metabolism of BMS-690514, a Potent Inhibitor of EGFR and VEGFR2. Journal of Pharmaceutical Sciences. 2010, 99(8),3579–3593.
2.Herbst RS."Review of epidermal growth factor receptor biology". Int. J. Radiat. Oncol. Biol. Phys.2004, 59 (2 Suppl): 21–6.
3.Yosef Yarden and Joseph Schlessinger. "Epidermal Growth-Factor Induces Rapid, Reversible Aggregation of the Purified Epidermal Growth-Factor Receptor". Biochemistry 1987, 26 (5): 1443–1451.
4.Downward J, Parker P, Waterfield MD. "Autophosphorylation sites on the epidermal growth factor receptor". Nature 1984, 311 (5985): 483–5.
5.Oda K, Matsuoka Y, Funahashi A, Kitano H. "A comprehensive pathway map of epidermal growth factor receptor signaling". Mol. Syst. Biol. 2005, 1 (1): 2005.0010.
6.Zhang H, Berezov A, Wang Q, Zhang G, Drebin J, Murali R, Greene MI. "ErbB receptors: from oncogenes to targeted cancer therapies". J. Clin. Invest. 2007, 117 (8): 2051–8.
7.Haizheng Hong. et al. Mechanistic Studies on a P450-Mediated Rearrangement of BMS-690514: Conversion of a Pyrrolotriazine to a Hydroxypyridotriazine.Chem. Res. Toxicol. 2011, 24, 125–134.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















