Oleanolic acid derivative 2 |
| Catalog No.GC36796 |
Oleanolic acid derivative 2 is an oleanolic acid derivative, which is a novel triterpenoid-steroid hybrid molecule.
Products are for research use only. Not for human use. We do not sell to patients.
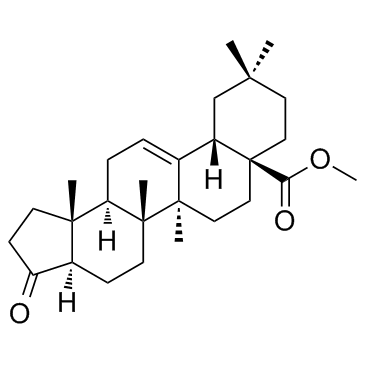
Cas No.: 211516-63-1
Sample solution is provided at 25 µL, 10mM.
Oleanolic acid derivative 2 is an oleanolic acid derivative, which is a novel triterpenoid-steroid hybrid molecule.
Oleanolic acid suppress transcription or translation of inducible nitric oxide synthase (iNOS) and inducible cyclooxygenase (COX-2) genes. extracted from the reference, compound 12a. For the transformation of olefin 9a into ketone 12a , the obvious ozonolysis cannot be used because ozone also reacts with the C-12 olefin of oleanane triterpenoids to give the 12R,13R-epoxide, 12-ketone,9 and 12R-hydroxy lactone.[1].
[1]. Tadashi Honda, et al. Design and Synthesis of 23,24-Dinoroleanolic Acid Derivatives, Novel Triterpenoid-Steroid Hybrid Molecules. J. Org. Chem., 1998, 63 (14), pp 4846-4849.
Average Rating: 5 (Based on Reviews and 16 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















