Letermovir (Synonyms: AIC246) |
| Catalog No.GC12672 |
A selective inhibitor of HCMV replication
Products are for research use only. Not for human use. We do not sell to patients.
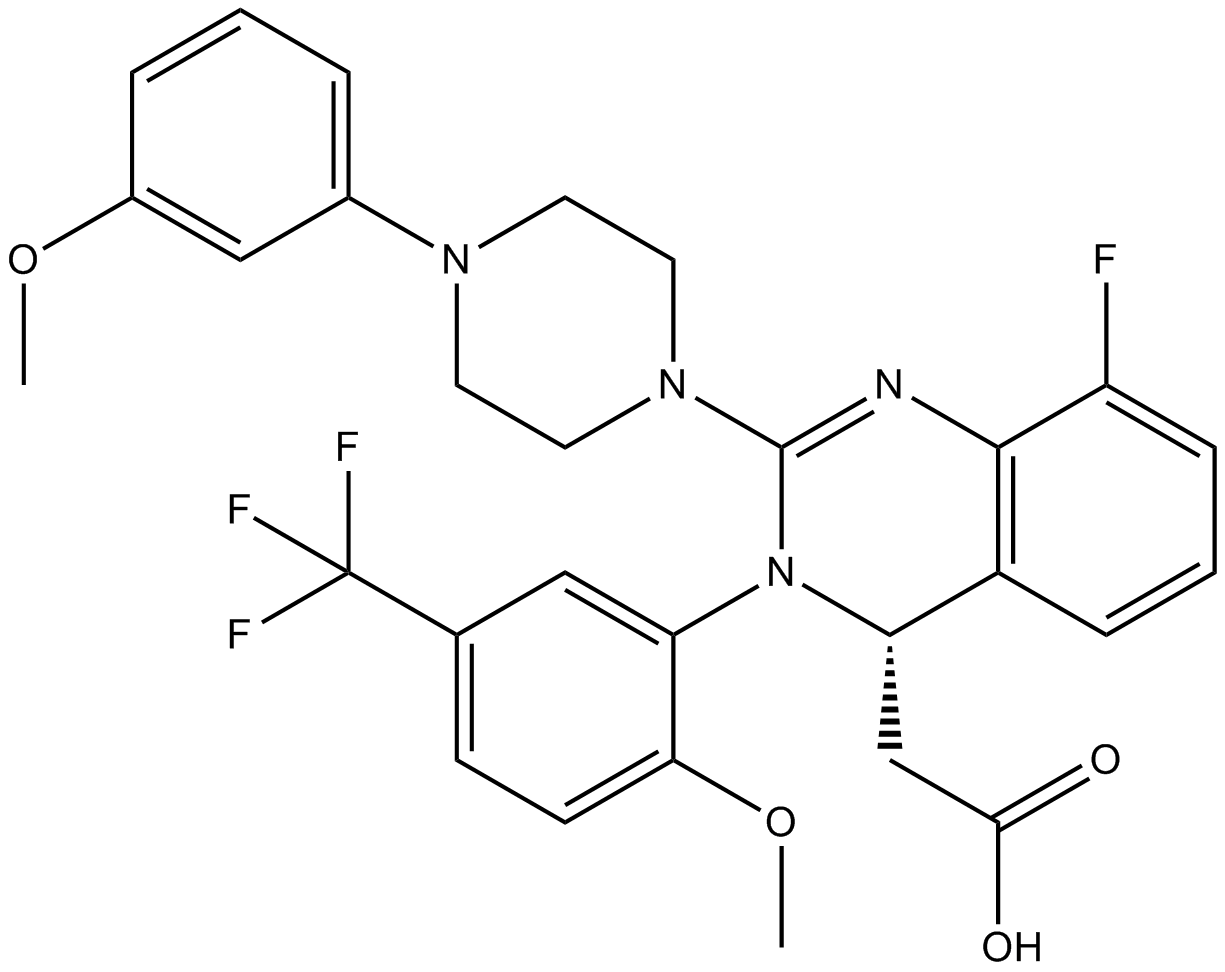
Cas No.: 917389-32-3
Sample solution is provided at 25 µL, 10mM.
AIC246, also known as letermovir, is a novel anti-CMV compound with IC50 value of 5.1 ± 1.2 nM. It targets the pUL56 (amino acid 230-370) subunit of the viral terminase complex [1].
The subunit pUL56 is a component of the terminase complex which is responsible for packaging unit length DNA into assembling virions.
AIC246 has a novel mode of action targets the enzyme UL56 terminase and keep active to other drug-resistant virus. The anti-HCMV activity of AIC246 was evaluated in vitro by using different HCMV laboratory strains, GCV-resistant viruses. The result showed that the inhibitory potentcy of AIC246 surpasses the current gold standard GCV by more than 400-fold with respect to EC50s (mean, ∼4.5 nM versus ∼2 μM) and by more than 2,000-fold with respect to EC90 values (mean, ∼6.1 nM versus ∼14.5 μM). In the CPE-RA strains, the EC50 values of AIC 246 ranged from 1.8 nM to 6.1 nM [2].
In mouse model with HCMV subcutaneous xenograft, oral administration of AIC246 caused significant a dose-dependent reduction of the HCMV titer. 30 mg/kg/d AIC246 for 9 days induced PFU reduction with maximum efficiency, compared with the gold standard GCV at the ED50 and ED90 level [2].
References:
[1].Verghese PS, Schleiss MR. Letermovir Treatment of Human Cytomegalovirus Infection Anti-infective Agent. Drugs Future. 2013, 38(5):291-298.
[2]. Lischka P1, Hewlett G, Wunberg T, et al.In vitro and in vivo activities of the novel anticytomegalovirus compound AIC246.Antimicrob Agents Chemother. 2010, 54(3):1290-1297.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




















