SBE-β-CD (Sulfobutylether-β-Cyclodextrin) |
| Catalog No.GC30001 |
SBE-β-CD (Sulfobutylether-β-Cyclodextrin) is a sulfobutylether β-cyclodextrin derivative used as an excipient or a formulating agent to increase the solubility of poorly soluble agents.
Products are for research use only. Not for human use. We do not sell to patients.
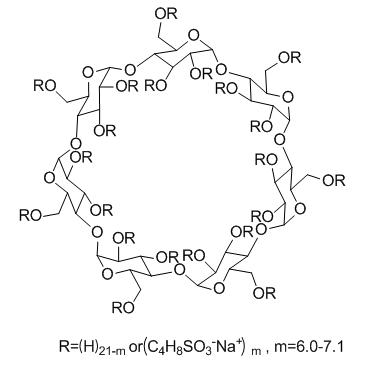
Cas No.: 182410-00-0
Sample solution is provided at 25 µL, 10mM.
SBE-β-CD, as a chemically modified β-CD, is a sulfobutylether β-cyclodextrin derivative. It always used as an excipient or a formulating agent to increase the solubility of poorly soluble agents.[1] SBE-β-CD is used for enhancing the aqueous solubility of lipophilic drugs. And SBE-β-CD is very safe.[2] Because of SBE-beta-CD has polyanionic nature, SBE-beta-CD interacts very well with neutral drugs to facilitate solubility and chemical stability.[3]
In vitro, the solubility of nimodipine in the presence of SBE-β-CD increased more than in the presence of HP-β-CD in distilled water. Nimodipine has more affinity because of the highest K1:1 value exhibited by SBE-β-CD,which be ascribed to the presence of the four-carbon butyl chain coupled with repulsion of the end group negative charges which allows for an “extension” of the hydrophobic region of the CD cavity.[4] At 400 mM SBE-β-CD concentration, the solubility of progesterone was ~7000-fold greater than its intrinsic solubility, aided by the formation of SBE-β-CD-progesterone complex. In addition, to prevent the displacement of progesterone from the complex by gastrointestinal contents, it needs to optimize the progesterone oral formulations by increasing the levels of SBE-β-CD in the formulation.[5]
In vivo efficecy test it shown that the SBE-β-CD (128 M−1) had a higher solubilizing ability than that of HP-β-CD (79 M−1) suggesting suggest that a simple mixing of SD-CS and SBE-β-CD can be potentially useful for the controlled release of a medicine for the continuous treatment of hypertension. In vivo absorption of the drug after oral administration to rats indicated the slow release characteristics of the SD-CS/SBE-β-CD composite in vitro.[6]
References:
[1].Fukuda M, et al. Influence of sulfobutyl ether beta-cyclodextrin (Captisol) on the dissolution properties of a poorly soluble drug from extrudates prepared by hot-melt extrusion. Int J Pharm. 2008 Feb 28;350(1-2):188-96.
[2].Parvathaneni V, et al. Repurposing Bedaquiline for Effective Non-Small Cell Lung Cancer (NSCLC) Therapy as Inhalable Cyclodextrin-Based Molecular Inclusion Complexes. Int J Mol Sci. 2021 Apr 30;22(9):4783.
[3].Stella VJ, He Q. Cyclodextrins. Toxicol Pathol. 2008 Jan;36(1):30-42.
[4].Semcheddine F, et al. Effects of the Preparation Method on the Formation of True Nimodipine SBE-β-CD/HP-β-CD Inclusion Complexes and Their Dissolution Rates Enhancement. AAPS PharmSciTech. 2015 Jun;16(3):704-15.
[5].Shankar VK, et al. Optimization of sulfobutyl-ether-β-cyclodextrin levels in oral formulations to enhance progesterone bioavailability. Int J Pharm. 2021 Mar 1;596:120212.
[6].Anraku M, et al. Design and Evaluation of An Extended-Release Olmesartan Tablet Using Chitosan/Cyclodextrin Composites. Pharmaceutics. 2019 Feb 15;11(2):82.
Average Rating: 5 (Based on Reviews and 4 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *



