Angeli’s Salt (Synonyms: Sodium α-oxyhyponitrite) |
| Catalog No.GC19428 |
A classical nitroxyl (NO-) donor
Products are for research use only. Not for human use. We do not sell to patients.
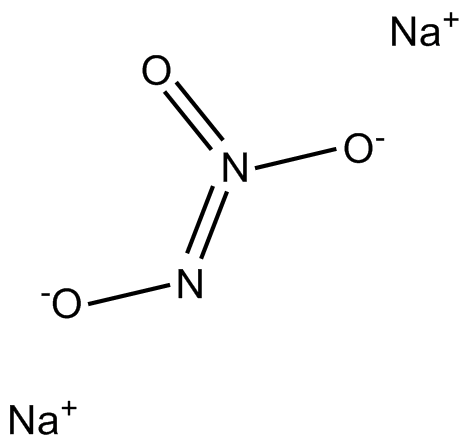
Cas No.: 13826-64-7
Sample solution is provided at 25 µL, 10mM.
Angeli’s salt is regarded as a classical nitroxyl (NO-) donor, but under certain conditions evolution of NO is also observed.1 It spontaneously dissociates in a pH-dependent, first-order process with a half-life of 2.3 minutes at 37°C (pH 7.4) to liberate 0.54 moles of NO per mole of parent compound.[1],[2]
Reference:
[1]. Maragos, C.M., Morley, D., Wink, D.A., et al. Complexes of ∙NO with nucleophiles as agents for the controlled biological release of nitric oxide. Vasorelaxant effects Journal of Medicinal Chemistry 34, 3242-3247 (1991).
[2]. Keefer, L.K., Nims, R.W., Davies, K.M., et al. “NONOates” (1-substituted diazen-1-ium-1,2-diolates) as nitric oxide donors: Convenient nitric oxide dosage forms Methods in Enzymology 268, 281-293 (1996).
Average Rating: 5 (Based on Reviews and 37 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




