Anastrozole (Synonyms: Anastrol, ICID 1033, ZD 1033) |
| Catalog No.GC10256 |
Aromatase inhibitor
Products are for research use only. Not for human use. We do not sell to patients.
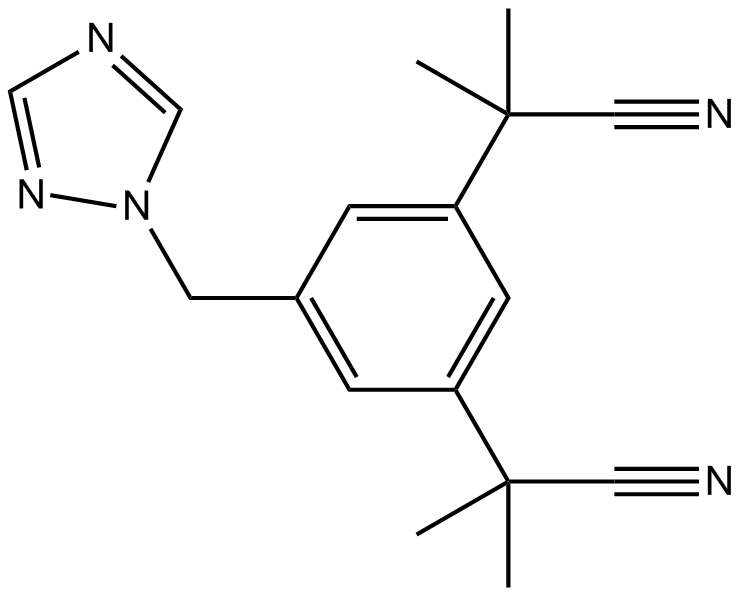
Cas No.: 120511-73-1
Sample solution is provided at 25 µL, 10mM.
Anastrozole (Arimidex,ZD1033) is a potent and selective inhibitor of aromatase with an IC50 value of 14.6 nM or 0.0043μg/ml [1].
Anastrozole has been reported to inhibit human placental aromatase with an IC50 value of 14.6 nM or 0.0043μg/ml. In addition, an oral concentration of 0.1mg/kg of anastrozole has been revealed to completely inhibit ovulation by given on day 2 or day 3 of the cycle. In immature rat, an oral concentration of 0.1mg/kg of anastrozole has also noted to completely extinguish the uterotrophic activity of exogenous AD. Apart from these, by guinea pig, dog and cow adrenal microsomes, anastrozole has been exhibited to suppress the conversion of 11-deoxycortisol to cortisol with mean IC50 values of 4.09μM,129μM and 11.9μM, respectively [1].
References:
[1] Dukes M1, Edwards PN, Large M, Smith IK, Boyle T. The preclinical pharmacology of "Arimidex" (anastrozole; ZD1033)--a potent, selective aromatase inhibitor. J Steroid Biochem Mol Biol. 1996 Jul;58(4):439-45.
Average Rating: 5 (Based on Reviews and 30 reference(s) in Google Scholar.)
GLPBIO products are for RESEARCH USE ONLY. Please make sure your review or question is research based.
Required fields are marked with *




